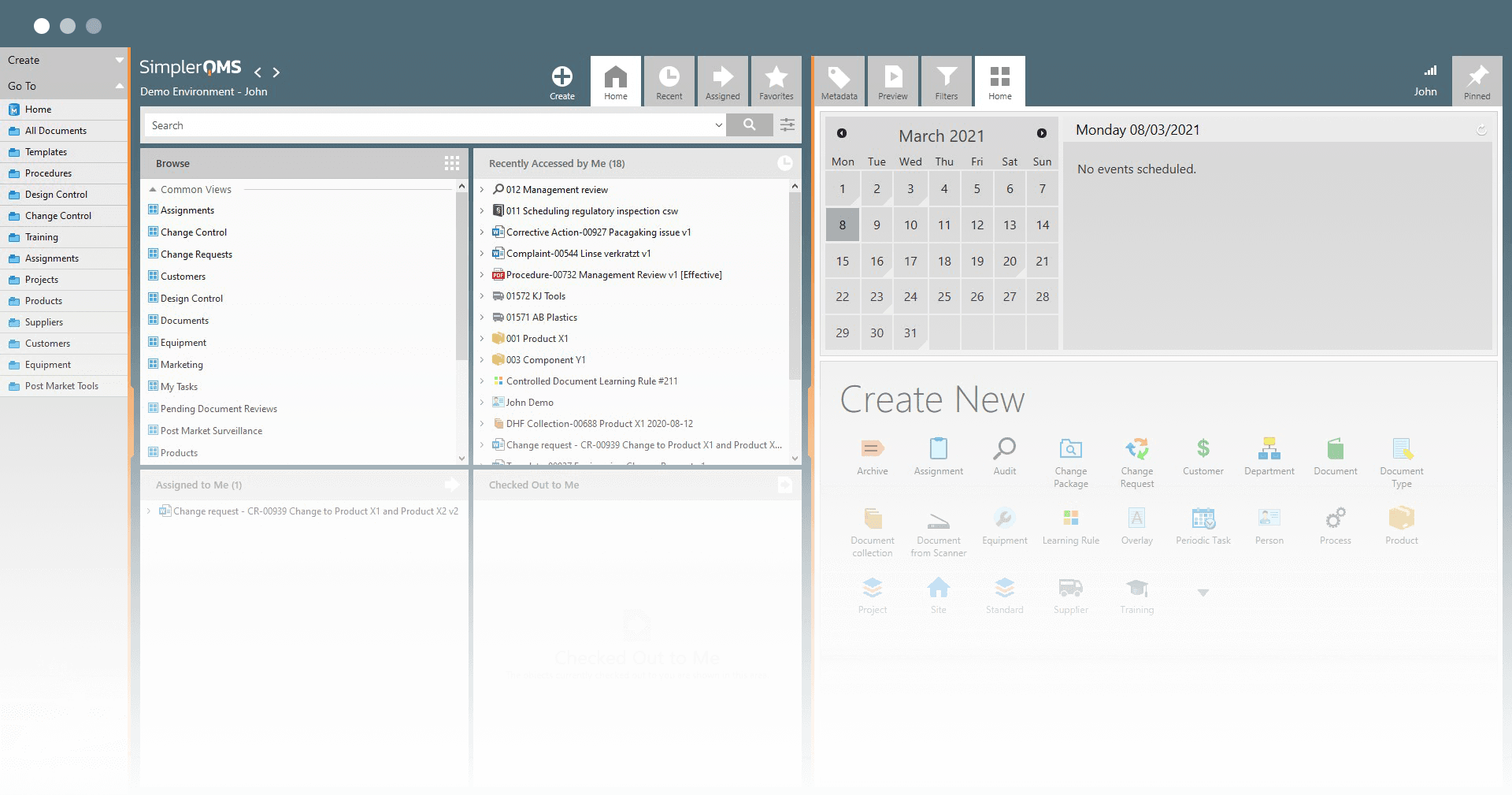
Calibration Management Software
Ensure high equipment quality, compliance, and boost your employee productivity by automating your equipment calibration processes.
Automate your equipment calibration activities
SimplerQMS equipment calibration software helps you in managing your calibration schedule and automatically triggers calibration tasks before the due date.
The system assigns calibration activities to the appropriate personnel and sends email notifications when it is time for them to take action.
Quickly access complete calibration history
With SimplerQMS, a complete equipment calibration log is readily available for audits and inspections.
All equipment-related records and approvals are stored in our secured cloud-based storage and can easily be found in a single place.
Ensure accuracy and minimize production downtime
Automate, route, and track all your calibration activities with ease. This can help you prevent and identify eventual failures. With SimplerQMS, you can easily escalate overdue assignments to supervisors and link failure incidents to specific equipment.
SimplerQMS allows you to administer specific instructions, such as measuring parameters, adjustment limits, and failure limits. You can then link these to specific work instructions for equipment calibration tasks.
Connect calibration management to other processes
SimplerQMS equipment calibration module is seamlessly integrated with other sub-systems, such as Electronic Batch Records (EBR) software. This allows you to maintain equipment-related documentation in a single location while staying in compliance.
Looking for more modules?
Regardless of the stage of your Life Science organization, our integrated software modules cover all your needs.
Training Management
Save time with automated training activities, learning overview, reminders, and generation of training certificates.
CAPA Management
Identify, uncover, resolve, and report all the preventative actions and corrective actions (CAPAs) seamlessly.
Complaint Management
Reduce the associated risks and resolve issues quickly by optimizing complaint management processes.
Change Management
Recognize and manage all changes accordingly to ensure compliance and structure within your organization’s QMS.
Design Control
Manage all the necessary processes related to product design and meet design control requirements with ease.
Document Control
Automate and standardize your document control activities with ease.
Issue Management
Simplify your issue management workflows by automating tedious tasks.
Electronic Signatures
Sign and send the necessary documents for authoring, review, approval, from anywhere in the world, at any time.
Product Lifecycle
Plan and organize all your product management activities and integrate them with other processes.
Audit Management
Automate your audit-related activities and ensure better audit preparedness.
Risk Management
Consolidate risk and handle your risk management file in a well-organized and structured manner.
Supplier Management
Simplify supplier-related activities and handle your supplier documentation following the standards.
Form Management
Standardize your documentation processes and boost employee productivity with effective document forms.
Deviation Management
Streamline the investigation, documentation, and resolution of all your deviations.
Electronic Batch Records
Digitize and automate your batch record processes to ensure regulatory compliance.
Nonconformance Management
Identify, evaluate, analyze and manage nonconformances more efficiently.
Trusted by companies around the world
























Ready to learn more?
To learn how you can make the most of SimplerQMS, request a free, personalized demo presentation.