Nonconformance Management Software for Life Sciences
Capture and address nonconformances to correct issues and support timely resolution.
Book a DemoJoin Over 5,000+ Quality Professionals Who Trust SimplerQMS
Nonconformance Management In a Complete eQMS
Nonconformance management software by SimplerQMS supports Life Science organizations in managing quality events such as nonconformances in a more streamlined manner.
SimplerQMS solution offers a document template package based on Life Science requirements, the capability to relate documents to relevant requirement chapters, select workflow participants, Issue Handlers and their responsibilities, set reminders before due dates, schedule tasks to assess the effectiveness of actions, and more.
SimplerQMS nonconformance management module is an integral part of our complete quality management solution, including all core Life Science modules such as training, CAPA, supplier, audit management, document control, and much more.

Identify, Analyze, and Manage Nonconformances
Create documents using a template package based on Life Science requirements and follow preconfigured workflows to manage nonconformances with ease.
Escalate nonconformance to CAPA with just a few clicks using our intuitive Document Action menu. Have a clear view of the document information, such as department, Issue Handlers, QA Responsible, due dates, and more, by accessing the metadata card.
Schedule assignments, tasks, and periodic checks to assess the effectiveness of actions related to nonconformances. Set up automatic notifications to remind the assigned people about the required activities before the due dates.
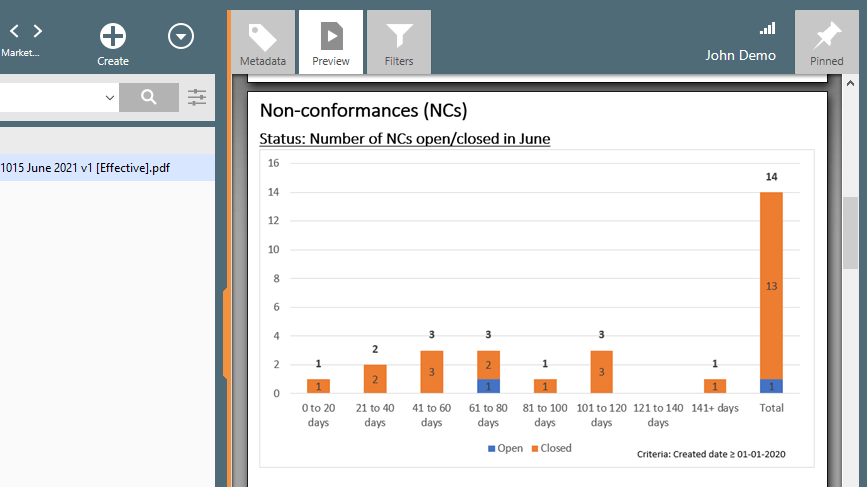
Easily Monitor the Status of Nonconformances
Monitor nonconformances by product, process, customer, equipment, supplier, or other data points using customizable views.
Keep track of all document changes. Every action is recorded in the system, providing a complete, time-stamped audit trail.
Easily search in the system for all nonconformance issues and overview their status.
Overview KPI reports for management input or export data from the views for further analysis if required.

Ensure Compliance With Life Science Requirements
Store all documents in a single cloud-based system. Search for documents by using the exact words mentioned in the title or content of the document. Easily retrieve documents, for example, during an audit situation.
SimplerQMS software supports compliance with Life Science requirements regarding post-market surveillance, including reporting to authorities, if necessary.
Electronically sign all documents using FDA 21 CFR Part 11 and EU Annex 11 compliant electronic signatures.
Connect All Customer Management and Quality Processes
Control all processes in one system with closed-loop workflows to address any nonconformity. All QMS modules, such as CAPA, change control, complaints, and more, are already included and interlinked in a complete eQMS software solution.
For example, a nonconformance could easily be escalated to a CAPA process and lead to document(s) being updated. After the document update, automatic notifications for retraining activities would be sent to the relevant people, all within a single system.
You can also utilize SimplerQMS with existing systems such as CRM, ERP, LMS, MES, and PLM to manage the entire product lifecycle.

What Customers Achieve With SimplerQMS
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
Discover How SimplerQMS Can Help You
Complete eQMS Software for Life Sciences
Frequently Asked Questions
Nonconformance Management Software is a solution that helps organizations to effectively manage nonconformances. It allows for tracking, documenting, and resolving nonconformances in a structured and organized way.
The software typically includes features for capturing, documenting, and managing nonconformances, escalating nonconformances to CAPAs, relating nonconformances to other documents, and sending automatic notifications to workflow participants regarding their tasks and assignments. It also provides visibility into the current state of nonconformance via highly configurable views.
SimplerQMS nonconformance management software is part of a complete QMS with all core life science modules included, such as document control, change management, training management, and more.
Nonconformance management software offers organizations the benefit of streamlining all activities related to resolving nonconformities, from creating a nonconformance document to escalating to a CAPA and checking the effectiveness of the actions performed, if necessary.
SimplerQMS provides a secure and cloud-based system where all processes are interlinked.
We also offer a complementary document template package based on Life Science requirements and preconfigured workflows that can help organizations achieve and maintain compliance.
Daily activities will be streamlined and simplified, such as relating documents, assigning Issue Handlers and QA Responsible people, searching and retrieving documents during audits, setting up automatic notifications, overviewing the status of processes, and more.
A nonconformance workflow in SimplerQMS starts with creating a document draft from one of the complementary templates or your own personalized template.
You can assign the Issue Handler and QA Responsible, determine the severity of the issue, set up a due date, relate the document to products and regulatory chapters, and more.
The software has built-in features to support reporting to regulatory authorities as a part of your nonconformance handling process, if necessary.
After the draft is completed, you can notify Issue Handlers, that will evaluate the need to escalate the nonconformance to a Corrective and Preventive Action (CAPA) and assess its effectiveness.
Yes, nonconformance software by SimplerQMS is compliant with FDA 21 CFR Part 11 and EU Annex 11.
SimplerQMS non-conformance management software is an integral part of an integrated solution. We offer all life science QMS modules, implementation, user training, and ongoing support included in one license at no additional cost.
The total cost will vary based on how many licenses you purchase.
Please visit our pricing page to learn more about the different license types, as well as the features and services included.
