QMS Software for Contract Research / Manufacturing Organizations (CRO / CMO)
Streamline operations and maintain regulatory compliance with purpose-built quality management software for Contract Research / Manufacturing Organizations (CRO / CMO).
Book a DemoJoin Over 5,000+ Quality Professionals Who Trust SimplerQMS
Ensure Regulatory Compliance in Your Contract Organization
Contract manufacturers, clinical research organizations, and testing laboratories (CxOs) must comply with the same regulatory requirements as their clients in the pharmaceutical, biotechnology, medical device, and other life science industries.
CxOs must maintain compliance with relevant standards, regulations, and regulatory guidelines in Life Sciences depending on the clients they serve such as GxP, FDA 21 CFR Part 11, 210, 211, and 820, ISO 13485:2016, ICH Q10, EU MDR and IVDR, EU GMP Annex 11, ISO 9001:2015, and others.
Compliance with these requirements can be achieved easier and faster with a purposefully built QMS software solution for Contract Organizations from SimplerQMS.
See certification & complianceOne Platform That Integrates All Quality Processes
With the SimplerQMS software solution, you can streamline regulatory compliance and typical CRO / CMO quality control processes by automating repetitive documentation activities.
Integrate all your quality assurance and control processes, store data in centralized, cloud storage, and manage risks, training, audits, suppliers, and other processes with ease.


Work More Efficiently in Microsoft Office Environment
SimplerQMS integrates with native Microsoft Office applications, including Word, Excel, Outlook, and PowerPoint, so you and your team can work with the tools you’re already familiar with.
Simply work on your documents within Microsoft Office applications and once the work is done, save them with one click in SimplerQMS Cloud. No manual download and upload of documents are required.
Streamline Your Document Control Processes
Automate document routing, follow-ups, tracking, escalation, review, approval, and other document-based processes.
With features like time-stamped audit trails and FDA 21 CFR Part 11 compliant electronic signatures built into the SimplerQMS solution, you can be confident that all documents are processed efficiently and follow the applicable regulatory requirements.
Learn more about document control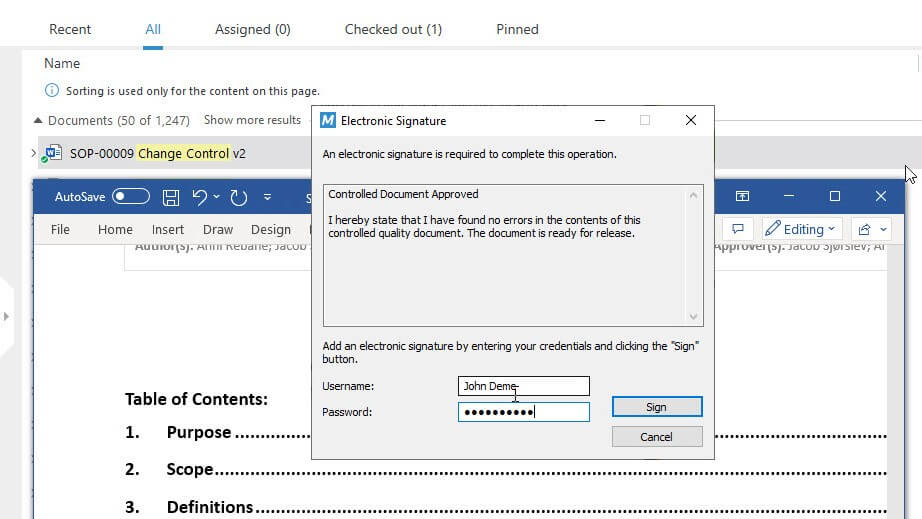

Forget About Time-Consuming Validation Activities
Software validation is a requirement for CROs and CMOs as well as other life science organizations that store and manage quality records electronically.
SimplerQMS provides a fully validated solution and performs monthly re-validation of the system, in compliance with FDA 21 CFR Part 11, ISO 13485:2016, and GxP guidelines. This means that your organization won’t have to spend time on system validation processes.
What Validation Documentation Evidence Does SimplerQMS Provide?
- Validation Procedure
- Validation Plan
- Validation Report
- IQ, OQ and PQ Documentation
Manage Nonconformance More Easily
The SimplerQMS nonconformance management solution facilitates the process of identifying, evaluating, analyzing, and managing non-conformances.
Access a single source of truth where all processes are connected from non-conformances and other quality events to training, audit findings, and more
Learn about non-conformance management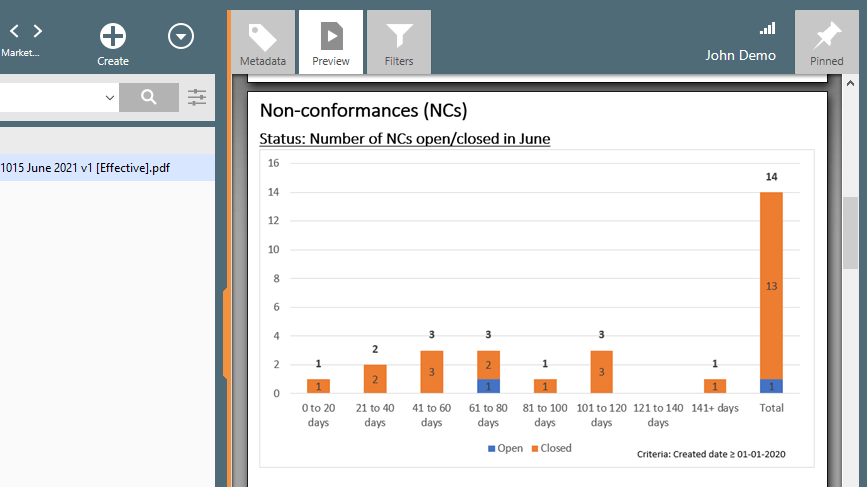

Resolve Customer Complaints Quickly and Effectively
As a CRO / CMO, it’s of the highest importance for you to meet the requirements of your main customers without delays and quickly resolve any complaints.
SimplerQMS provides robust customer complaint-handling capabilities which allow you to track, manage, analyze and resolve customer complaints in a timely and effective manner.
Explore complaint management capabilitiesAutomate and Gain Traceability of Your CAPA Processes
The SimplerQMS CAPA management software solution integrates with different QMS sub-systems and provides a closed-loop CAPA process.
Save time with automated CAPA workflows and pre-configured CAPA forms, and avoid any errors in manual data entry or process execution.
Learn more about CAPA management

Manage Changes More Effectively
The change control module by SimplerQMS allows you to automatically track changes, collect data, and send changes through an approval process.
Automated follow-ups, notifications, and escalation activities help you quicker finalize changes to SOPs in your system.
Explore our change control moduleExpedite Employee Training Management Processes
With continuous changes taking place daily, you must ensure that employee training in your contract organization is well-documented and in compliance with regulatory requirements.
SimplerQMS training management module streamlines training processes by automating the assignment of training activities, reminders, generation of training certificates, and more.
Explore training management features
All-in-One QMS Software for Life Sciences
View all modulesFrequently Asked Questions
As a Contract Research / Manufacturing Organization (CRO / CMO), you are responsible for ensuring that the services you provide meet customer expectations and regulatory requirements. An effective quality management system (QMS) is essential to help you manage your processes and ensure compliance. By implementing a QMS, you can streamline your operations, improve communication and collaboration between different departments, and ultimately provide better quality services to your customers.
QMS software solutions offer many benefits for Contract Organizations, including streamlined operations and improved efficiencies, reduced paper documentation and associated costs, increased visibility into quality data for better decision making, improved compliance with regulatory requirements, and enhanced communication between sites, sponsors, and staff.
Yes, SimplerQMS provides a fully validated software solution. It is validated according to ISPE GAMP5 and undergoes revalidation for each new version or standard update.
We do all the software validation for you – there are no additional expenses, resources, or time commitments to software validation on your part.
SimplerQMS complies with requirements regarding the validation of systems used for QMS such as 21 CFR Part 11, and 820, EU GMP Annex 11, and ISO 13485:2016.
Furthermore, we provide extensive validation evidence for use during audits and inspections, including the validation plan, procedures, and reports, as well as IQ, OQ, and PQ documentation.
SimplerQMS provides an all-in-one QMS software solution that includes all modules, implementation, training, ongoing support, validation, hosting, and more. With SimplerQMS, you pay one price that covers everything – there are no other subscription fees.
The exact price of the solution differs depending on how many people will use it, and the license types required. Therefore we recommend you book a tailored demo and talk to our experts to request a pricing quote for the needs of your organization.
Depending on whether you already have a quality management system (QMS), and the number of documents, implementation time may be shorter or longer. Our experience shows that the average implementation time for a new QMS is between five and six weeks. However, we have seen clients go live in as little as three weeks.
Our team of experts will work with you every step of the way to ensure a smooth and successful transition to our software.
What Customers Achieve With SimplerQMS
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
What Customers in the Pharmaceutical and Biotechnology Industries Say
“User-friendly and simple to use. Easy to customize to fit your individual needs. The modular system ensures that users can only see relevant modules.”
“Exceptional customer service, excellent “hands-on” training, and they are willing to help my team integrate other applications with SimplerQMS. They have been true partners in implementing SimplerQMS.”
“Simple system that was incredibly easy to set up. It comes pre-validated, which, in turn, saves the company a lot of time and effort. Great customer support.”
See SimplerQMS in Action
To see SimplerQMS in action and learn how you can make the most of it, request a personalized demo presentation.
Book a Demo