21 CFR Part 11 Compliant Electronic Signature Software
Apply electronic signatures designed to support FDA 21 CFR Part 11 requirements for controlled approvals, traceability, and accountability.
Book a DemoJoin Over 5,000+ Quality Professionals Who Trust SimplerQMS
21 CFR Part 11 Compliant Electronic Signature Software Within a Complete eQMS
Title 21 CFR Part 11 is part of Title 21 of the Code of Federal Regulations that specify the requirements for electronic records and electronic signatures to ensure trust, reliability, and equivalence to paper records and handwritten signatures.
SimplerQMS complies with 21 CFR Part 11 by providing user authentication and access control, unique electronic signatures, linking each electronic signature to the corresponding document, time-stamped audit trails, etc.
In addition to Electronic Signatures, SimplerQMS offers a comprehensive eQMS software solution that covers all Life Science QMS modules. This includes document control, change management, training management, complaints management, nonconformance management, CAPA management, audit management, and other modules.
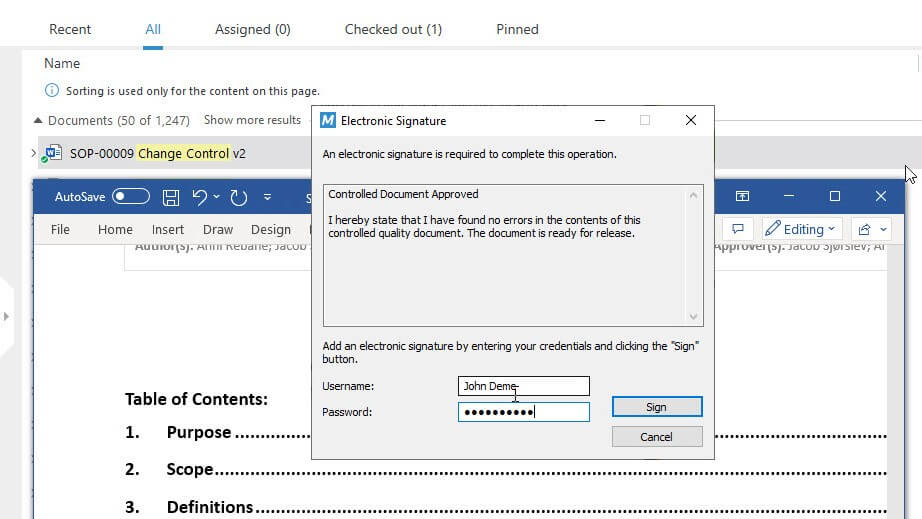
Sign Documents with 21 CFR Part 11 Compliant Electronic Signatures
Sign off documents using 21 CFR Part 11 compliant electronic signatures and ensure electronic records’ authenticity, integrity, and confidentiality.
Ensure user authentication with unique user logins and passwords. The system prevents users from having identical passwords and avoids any other individual’s reuse or reassignment of passwords.
Electronically sign documents and have the system automatically record the signer’s name, date, time, and signature justification as per 21 CFR Part 11.
Streamline and Automate Workflows to Improve Efficiency
Automate documentation processes, such as document routing for review or approval, and sending of assignments, notifications, and reminders, for faster document processing times and approvals.
Use forms and document templates to help ensure consistency and accuracy while creating documents.
Minimize human error and reduce manual processes by streamlining and automating your document workflows with SimplerQMS.


Streamline Your Work with Microsoft Office Integration
Use the familiar Microsoft Office applications to edit and collaborate on documents.
Work on your documents in Microsoft Word, Excel, and PowerPoint applications.
Once the work is done – save the documents with a single click to the cloud, eliminating the need for manual document downloads and uploads.
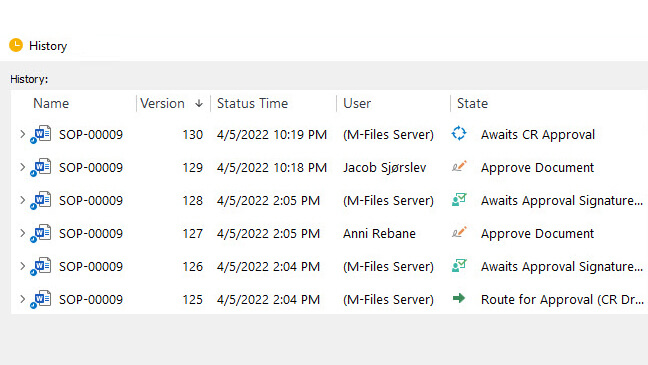
Track Every Action with Robust Audit Trails
Automatically record and track all actions made to the documents. The system automatically creates a time-stamped audit trail and improves the traceability of information.
Ensure the authenticity of electronic signatures with a system that automatically links them to the respective electronic records.

Control User Access and Validate User Identity
Limit information access only to authorized personnel by providing user access to the system based on job roles or only to specific employees.
Improve data security by employing procedures and controls designed to ensure data privacy and confidentiality.
Control access to the system using Microsoft Entra ID (previously known as Microsoft Azure Active Directory) or Google Workspace SSO and ensure only authorized personnel can access the system.
Keep Comprehensive Records and Maintain Version Control
Automatically record all changes made to documents and retain the electronic records of all versions for the required period.
Maintain a history of all document versions and have the ability to compare changes easily and roll back to older versions if necessary.
Facilitate document retrieval by utilizing a search function that enables users to search keywords in document titles and content.

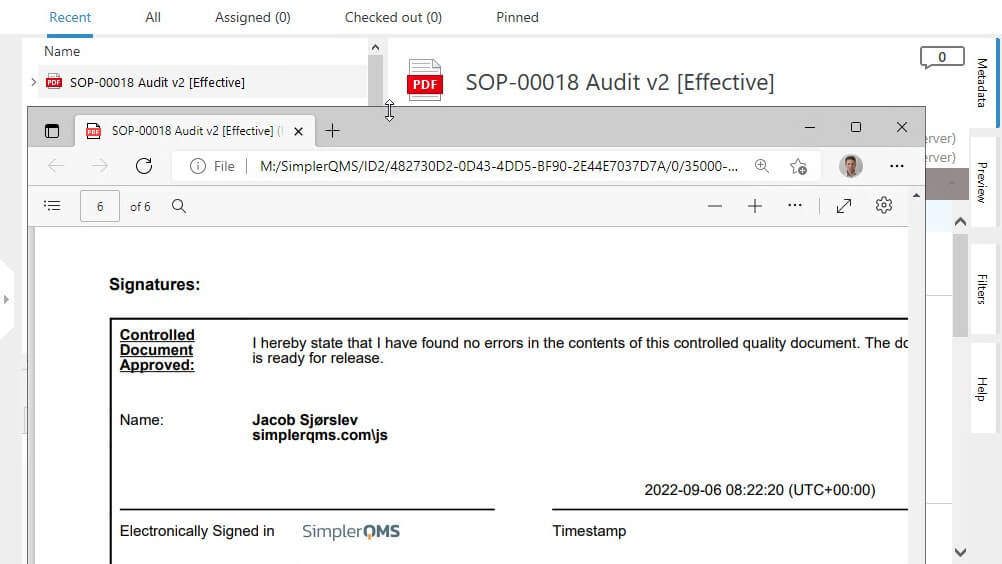
Sign External Documents with Ease
Upload external documents into the system using the drag-and-drop feature.
Easily and securely sign off external documents using electronic signatures.
Share approved, signed, and released documents with external parties and improve collaboration.
Perform Documentation Tasks On-the-Go
Take advantage of a cloud-based system to perform documentation tasks remotely or on-the-go.
Increase productivity by eliminating the need to be in the office.
Review, approve, and sign off documents from any device, from anywhere, at any time.


Avoid Spending Time On System Validation Activities
Save time and resources by using a validated software solution.
SimplerQMS is fully validated according to ISPE GAMP5. The system is automatically re-validated every time a new version is released, or upon applying standard updates.
SimplerQMS software complies with Life Science-related requirements regarding validation, including 21 CFR Part 11, EU GMP Annex 11, 21 CFR Part 820, and ISO 13485:2016.
See Electronic Signatures and Workflows in Action
This short video showcases how SimplerQMS enables you to sign off any electronic record with our 21 CFR Part 11 compliant electronic signatures.
Automated workflows and notifications facilitate a seamless and fully digitized process that benefits internal and external users.
What Customers Achieve With SimplerQMS
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
Discover How SimplerQMS Can Help You
Much More Than Just Electronic Signatures
Frequently Asked Questions
Electronic signatures are 21 CFR Part 11 compliant if they comply with the requirements specified in the regulation.
Some requirements include ensuring that electronic signatures are unique to the individual, cannot be reused, and are linked to electronic records. Electronic signatures must be trustworthy, reliable, and equivalent to handwritten signatures.
Software validation for 21 CFR Part 11 involves following the test scripts to verify that the system functions as intended.
This entire process is documented according to the Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) procedures.
To better understand the advantages of a fully validated QMS software system, when validation is necessary, and how it is performed, check out our article on QMS software validation.
However, companies that choose SimplerQMS as their eQMS software do not need to worry about system validation.
Our system is fully validated, and we perform continuous re-validation to ensure ongoing compliance with Life Science requirements regarding the validation of computer systems.
SimplerQMS solution allows for electronic signatures on all documents that go through an approval workflow.
This means that any document that requires approval, such as change requests and standard operating procedures, can be signed off using electronic signatures.
However, not all documents need to be signed. Attachments, such as training certificates or other uncontrolled documents, do not have to need to be signed.
The 21 CFR Part 11 compliant electronic signatures are integral to the complete eQMS software solution by SimplerQMS and cannot be purchased as a separate product or feature.
The total cost depends on the number and type of licenses you acquire.
All Life Science QMS modules such as document control, change management, training management, supplier management, and so on, and included in the subscription price.
Furthermore, hosting, system validation, implementation, user training, and ongoing support are also all included in the subscription.
To learn more about SimplerQMS pricing and all the features and services included, visit our pricing page.
