Design Control Software for Medical Devices
Control and document design activities to manage risk and meet regulatory requirements.
Book a DemoJoin Over 5,000+ Quality Professionals Who Trust SimplerQMS
Design Control Software Built-Into an All-In-One eQMS
Medical devices must be designed in ways to ensure performance and reduce risk. Design Control Software by SimplerQMS helps you accomplish both and much more.
The Design Control Solution is just a part of an all-in-one eQMS system.
The SimplerQMS solution includes essential quality system modules such as change management, audit management, supplier management, non-conformance/deviation management, CAPA management, and beyond. All to support you in achieving compliance and placing your device in the market faster.

Create and Overview Your Design Control Documents
With SimplerQMS you will be able to overview and manage multiple Technical Files, Device History Records (DHR), Design History Files (DHF), and Device Master Records (DMR).
Use Word and Excel Templates to create new documents, drop external documents inside the system and migrate all existing files to a centralized cloud-based repository to document the design process. Utilize Template & Form Management features to help you easily create documents and enforce a standardized approach that helps comply with relevant requirements.
Use dashboards to overview design control documentation for products, projects, regulatory requirements, and so on, by utilizing SimplerQMS’ intuitive metadata card. You can also link documents to different design control archives such as TF, DHF, DHR, and DMR without creating duplicates.
Streamline Audit Process and Submission Handling
Simplify audits by quickly retrieving essential documents from the Design Control Software using dashboards and/or an internal search tool.
Use the document collection tool to compile technical documentation, Design History Files, Device Master Records, and Device History Records. And securely store all the needed documentation for each product that you would need for an EU MDR evaluation or 510(k) submission.
SimplerQMS also allows documents and collections to be exported to the local storage and shared with different partners.

Out-of-the-Box Regulatory Compliance
SimplerQMS supports medical device manufacturers to achieve compliance with regulations, including EU MDR and IVDR, 21 CFR Part 11 and Part 820, ISO 13485:2016, GxP, and others.
Our software is fully validated according to GAMP5. We perform monthly re-validation of the system, so you do not need to spend time on software validation activities.
The system complies with ISO 13485:2016, 21 CFR Part 820, 21 CFR Part 11, and the EU Annex 11 regarding validation and electronic signatures.
Communicate Effectively With Your Collaborating Partners
QMS paper-based systems can make it difficult for medical device manufacturers to share important documents with material suppliers, software providers, consultants, external auditors, and other third parties.
SimplerQMS digital solution allows companies to relate collaborators to specific sites and departments using metadata when creating documents. Give location-specific access to documents to communicate new and updated files.
Easily export documents from the Design Control Software to a local computer to share with collaborating partners if needed.


Organize Design Documentation Archives Related to Submission Requirements
Create archives to organize all documents required for regulatory submissions, whether EU MDR evaluation or 510(k) submission. The same document can be related to more than one collection without duplicates.
Restrict access to sensible files by assigning team members to specific project archives with intuitive metadata cards.
Manage your device Design Documentation for unique components and parts with the help of document templates.
Handle Risk Assessments
Connect SimplerQMS Design Control and Risk Management QMS modules to ensure your medical device is safe for use. Assessing risk while you create your design documentation can help you recognize and prevent issues from happening.
Capture and document all risk management processes by using built-in templates that can help streamline risk assessment activities. Support change in processes or products by relating risk documents to design control files, if necessary.
Ensure your device complies with standards such as ISO 14971:2019 and ISO 13485:2016 while in the design and development phase to optimize time to market and reduce complaints and non-conformances.


Centralized Cloud-Based Software Solution
Physical file storage in paper-based and hybrid systems can lead to misplaced or lost documents.
Ensure easy document access and traceability using a centralized and cloud-based system. Get access to documents in the cloud on your computer or mobile device from anywhere.
SimplerQMS Software uses Amazon Web Services (AWS) Cloud Data Centers to store data in one secure digital location. Have your documents stored in a reliable and secure system compliant with ISO 27001:2022 and 21 CFR Part 11.
Easily Record All Changes in Design Documentation
Use forms and templates to help keep track of design verification and validation, design reviews, and changes in documents.
SimplerQMS Design Control Software records all changes made in the documentation and provides time-stamped audit trails, revision history, and complete traceability. Document versioning features ensure documents are always up-to-date.
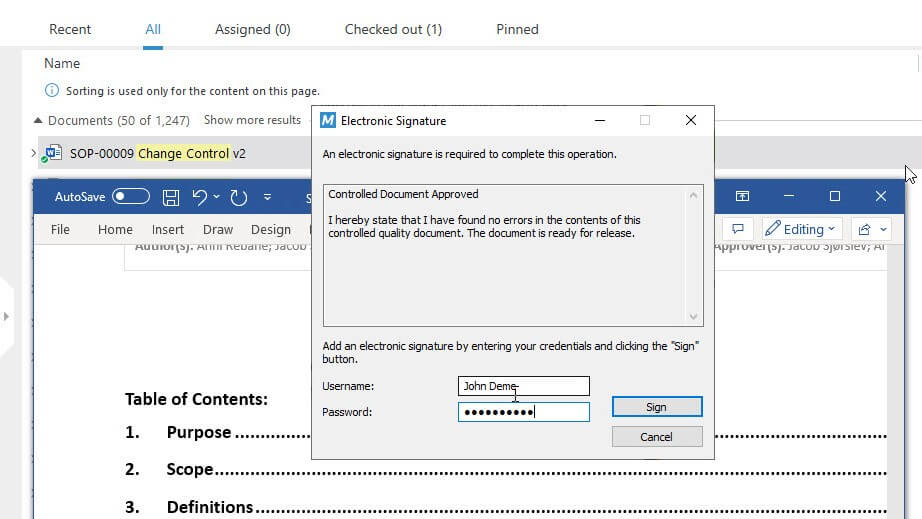
Keep track of changes even after design freezes with our Change Management Software. Create change request forms using templates and quickly assign tasks. After completion, sign documents electronically and enforce learning activities.
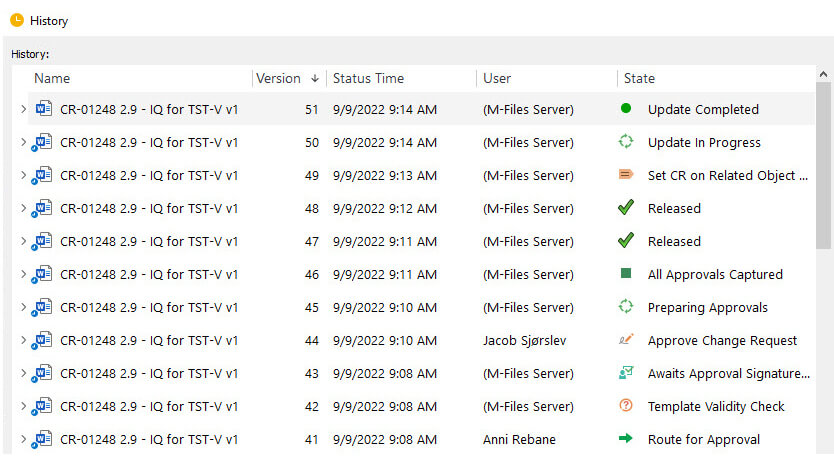

Ensure Adequate Competencies in Your Design Team
Product development time can be affected negatively by incomplete and incorrect documents. Avoid delays in documentation processes with a well-trained and up-to-date team.
Use the SimplerQMS training management capabilities to help check and improve your colleagues’ competencies by creating learning rules and personalized assignments. You can use several training materials such as videos, Word documents, PDFs, Excel tables, PowerPoint slides, quizzes, and more.
Develop Design Control SOPs to enforce learning activities using built-in templates. Maintain appropriate records of education to ensure compliance with regulations and standards.
Software Integration With Other Applications
Easily integrate project management, bug tracking, and other software solutions to SimplerQMS solutions via add-ons and API.
Incorporate documents from Jira Software to connect your technical documentation to the eQMS system while maintaining the traceability of all Design Control files.

See Our Design Control Possibilities in Action
Find out how SimplerQMS Design Control Software helps you create documents using templates, relate files with metadata, assign documents to multiple archives, export files, create snapshots of collections, and much more.
What Customers Achieve With SimplerQMS
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
Discover How SimplerQMS Can Help You
All-in-One QMS Software for Life Sciences
Frequently Asked Questions
Design control software allows medical device manufacturers to create, organize, and maintain the order of all technical files related to medical device development. These documents represent the history of a device’s development and are mandatory documentation to comply with regulations and standards.
By automating manual processes, the system speeds up product design and development cycles to ensure that products are released quickly and efficiently.
Design Control Software is just one part of a much bigger solution provided by SimplerQMS. We offer complete medical device eQMS software. Our solution provides many other core quality management system modules, such as audit management, CAPA management, equipment management, personnel training management, and much more.
Design controls are the way for medical device manufacturers to document and prove that a device is safe and effective to be placed on the US market through a 510(k) premarket submission.
SimplerQMS solution supports companies to achieve compliance with requirements to a 510(k) submission, 21 CFR Part 820, 21 CFR Part 11, and other relevant regulations.
Although SimplerQMS provides a helpful template package for document management, medical device manufacturers are still responsible for ensuring that the content of documents is compliant.
SimplerQMS solution helps to manage the mandatory design and development documentation as per ISO 13485:2016 and EU MDR.
You can create document collections, quickly search and retrieve documents, export files to your local storage, and organize documents for audits and regulatory submissions.
Our software is validated according to GAMP5. And it complies with ISO 13485:2016, 21 CFR Part 820, 21 CFR Part 11, and EU GMP Annex 11 regarding electronic signatures and validation.
Feel free to use our templates as an inspiration but remember that you are responsible for ensuring that the content of documents is compliant.
SimplerQMS Design Control solution is part of an all-in-one solution for eQMS.
Our solution includes all core QMS modules, implementation, user training, and ongoing support. Therefore, the total cost will vary based on how many licenses you acquire.
Please visit our QMS solution pricing page and learn more about our plans.
The time to implement SimplerQMS eQMS Software with built-in Design Controls depends on the volume of data that needs to be migrated or documents created within the software as well as the available resources within your organization.
Usually, it takes 5 to 6 weeks to implement the SimplerQMS solution.
