Document control is the systematic process used to control documents throughout their lifecycle. The document control process involves establishing procedures for reviewing, approving, tracking, and versioning documents. This process ensures that only the most recent document versions are used.
Document control is essential for ensuring compliance with standards and regulations, such as ISO 9001:2015, ISO 13485:2016, and FDA 21 CFR Parts 211, 212, and 820, among others.
Document control and document management are interconnected but different aspects of handling documents in a company. Document control is a part of document management.
Companies can implement a document control system to handle document control processes. A document control system is a structured set of processes and procedures designed to ensure control of documents.
The difference between a document management system and a document control system lies in the specific focus. A document management system allows users to easily find, share, and create new document versions. A document control system ensures that only the latest approved version of a document is accessible and outdated versions are archived.
This article focuses on the document control part of a document management system, which we will refer to as a document control system.
Document control in an electronic Quality Management System (eQMS) ensures that documents are properly reviewed, approved, tracked, and controlled to comply with relevant requirements.
SimplerQMS provides an eQMS solution designed for life science companies with robust document control capabilities. Book a personalized demo to experience how SimplerQMS can streamline your document control process.
What Is Document Control?
Document control is the systematic process of controlling documents and records throughout their lifecycle. Document control involves reviewing, approving, tracking, submitting, and versioning documents.
Document control ensures that documents, such as quality manuals, policies, procedures, and records, are properly maintained, secure, and accessible to authorized personnel.
The document control process ensures that only the most recent versions of documents are in use by controlling the document versioning and approval process.
Why Is Document Control Important?
Document control is a requirement in several regulations, standards, and guidelines across various industries.
Document control is important for ensuring consistency, traceability, accountability, and transparency in the handling of documents. Document control provides a structured process for controlling documents and helps achieve and maintain compliance with relevant requirements.
By systematically controlling documents, companies can prevent issues related to using outdated documents and ensure that all personnel have access to the most recent information.
What Is the Document Control Process?
The document control process involves a set of steps for controlling documents through their lifecycle.
Listed below are the main document control process steps.
- Document Identification: Assign a unique document identification number and version number. Ensure consistent formatting for titles and metadata.
- Document Review: Send the document to relevant personnel for review.
- Document Approval: Submit the reviewed document for approval by authorized personnel, recording approval details such as the date and approver’s information.
- Document Access: Store documents in a centralized, secure repository and manage permissions to ensure only authorized personnel can access them.
- Document Tracking: Maintain records of document changes and versions to track document history and status.
- Document Retirement: Remove obsolete or outdated documents from use. Mark retired documents to prevent accidental use.
The illustration below presents a diagram of the document control process.
What Is a Document Control System?
A document control system involves a set of processes and procedures designed to control documents throughout their lifecycle. The document control system ensures that documents are properly identified, reviewed, approved, versioned, and tracked.
A document control system includes processes for maintaining document security, managing version control, and overseeing document review, approval, and submission.
The document control system helps employees stay aware of the document’s state and ensures that only the latest approved document version is accessible while outdated versions are archived.
Document control systems can be paper-based, hybrid (combining paper and digital formats), or fully electronic, depending on the company’s needs and the resources available.
What Is the Difference Between a Document Control System and a Document Management System?
The main difference between a document control system and a document management system lies in its focus.
The document control system controls the review, approval, tracking, and versioning of controlled documents. A document control system ensures only the most recent approved versions of documents are accessible and archives outdated versions. It helps employees know the current state of each document.
The document management system provides functionalities for storing, managing, and distributing documents across a company. A document management system helps users create, find, and share the latest document versions.
Both document management and document control systems are essential and interconnected. A controlled document is a managed document, but not all managed documents are controlled. A controlled document cannot exist without practicing document management.
The illustration below presents the main differences between document control and document management systems.
What Is the Role of a Document Control System Within a Quality Management System (QMS)?
The role of a document control system within a Quality Management System (QMS) is to ensure that all documents are properly identified, reviewed, approved, and tracked. The document control system ensures that documents are handled in compliance with relevant requirements.
The document control system is essential in managing QMS documentation. QMS documentation includes documents and records that outline and support a company’s quality management processes, regulatory compliance, and continuous improvement efforts.
What Are the Requirements for Document Control in a Document Management System?
The requirements related to document control within the document management system are outlined in standards, regulations, and guidelines.
The following list includes some standards, regulations, and guidelines for QMS in the life science sector that outline the requirements for document control processes.
- ISO 9001:2015 (Quality Management Systems Requirements)
- ISO 13485:2016 (Medical Devices Quality Management Systems)
- ISO 15189:2022 (Requirements for Quality and Competence of Medical Laboratories)
- 21 CFR Part 820 (Quality System Regulation – QSR)
- EudraLex Volume 4 Good Manufacturing Practice (GMP) Part I
- ICH Q7 (Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients)
Below is a brief description of each requirement, highlighting their document control specifications.
NOTE
Please be aware that the information provided here is for educational purposes only. Companies must always refer to the official information provided by regulatory authorities to ensure compliance.
ISO 9001:2015
ISO 9001:2015 is an international standard that specifies the requirements for a quality management system.
Control of documented information in Section 7.5.3 requires documents to be controlled to ensure they are available, suitable, and protected from loss of integrity or misuse. Section 7.5.3 includes requirements for how documents are distributed, accessed, retrieved, stored, preserved, changed, and disposed of.
In addition, any external documents needed for the QMS must be properly identified and controlled. Documents must be kept as proof of compliance and protected from accidental changes.
ISO 13485:2016
ISO 13485:2016 is an international standard that specifies requirements for a quality management system in the medical devices industry.
Section 4.2.4 specifies that all documents needed for the QMS must be controlled. The control of documents includes reviewing and approving documents before they are used, updating and re-approving them as needed, and keeping track of their current versions and changes. Section 4.2.4 also specifies ensuring that the correct versions are available where needed, among other requirements.
Control of records in Section 4.2.5 requires that records must be maintained to provide evidence of conformity and effective QMS operation. Procedures must be in place to control the identification, storage, security, retrieval, retention, and disposal of records.
ISO 15189:2022
ISO 15189:2022 is an international standard that specifies requirements for quality and competence in medical laboratories to ensure consistent, reliable, and accurate test results for patient care.
Regarding specific guidelines for document control, Section 8.3 requires laboratories to control internal and external documents relevant to their operations. Documents must be uniquely identified, approved by authorized personnel, periodically reviewed, updated, and made available at points of use.
Section 8.4 on control of records requires laboratories to create and maintain legible records. Laboratories must have procedures for the identification, storage, protection, backup, archiving, retrieval, retention, and disposal of records, specifying retention times.
21 CFR Part 820
21 CFR Part 820 is an FDA regulation that outlines the quality system regulations for medical devices.
Section 820.40 requires documents to be controlled through written procedures. According to Section 820.40, document control procedures should ensure that documents are adequately reviewed, approved, distributed, and updated and that changes are documented.
EudraLex Volume 4 GMP Part I
EudraLex Volume 4 GMP Part I is a European guideline that contains guidance on the good manufacturing practices for medicinal products used in human and veterinary medicine.
Chapter 4, sections 4.1 to 4.6 on generation and control of documentation require all types of documents to be clearly defined and controlled.
Documents must be designed, prepared, reviewed, and have a unique identification. Documents should not be handwritten except for the space where data entry is necessary. Proper controls must be in place to maintain the integrity of documents throughout the document retention period.
ICH Q7
ICH Q7 is an international guideline that provides guidelines for good manufacturing practices for active pharmaceutical ingredients.
Section 6 specifies that all documentation and records approval, review, update, and disposal should be controlled and documented with revision histories.
What Types of Documents Need To Be Controlled?
Document control handles a variety of documents depending on specific industry requirements and company policies.
Documents typically controlled under a quality management system include manuals, policies, standard operating procedures (SOPs), specifications, forms, templates, and records, as described below.
- Quality Manuals: Outline the company’s quality system and its compliance with relevant requirements.
- Quality Policy and Objectives: Define the company’s commitment to quality and its specific goals.
- Standard Operating Procedures (SOPs): Provide directions on how tasks and processes should be carried out within a company.
- Specifications: Describe in detail the requirements that products or materials used or made during manufacture must comply with. Specifications serve as a basis for quality evaluation.
- Forms and Templates: Standardized documents used to consistently capture data and information and support document creation.
- Records: Provide evidence of completed actions in accordance with procedures, work instructions, and specifications.
These examples represent documents across various industries that need to be controlled to maintain quality and compliance.
What Are the Key Components of Effective Document Control?
Listed below are the key components of a document control system.
- Document Number
- Version Control
- Audit Trail
- Periodic Review of Documents
- Access Control
- Change Control
The implementation of these components, along with document approval, updates, and control of external documents, is detailed in document control SOPs and work instructions.
Document Number
The document number is a unique identifier assigned to each document. The document number is typically created by using a numbering system that combines numbers, letters, and sometimes dates.
The document number ensures easy tracking, retrieval, and identification of documents within the system through the document’s unique identification (ID) number.
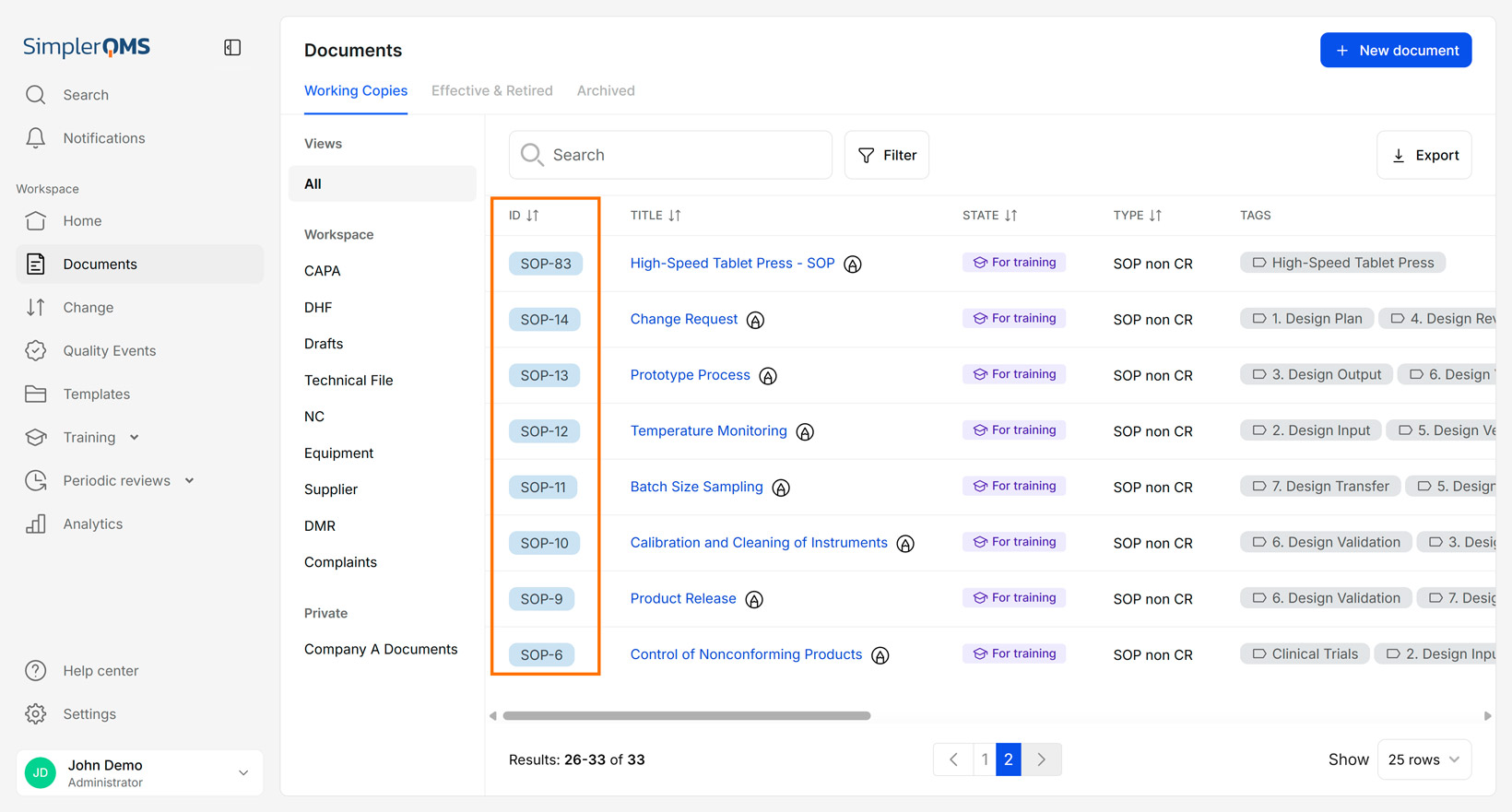
Version Control
Version control is the process of tracking document versions, ensuring that only the most recent version is available and clearly identified. Document versions are recorded in the revision history, providing transparency and accountability.
Version control prevents the use of outdated or incorrect documents, which can lead to compliance issues or operational errors.
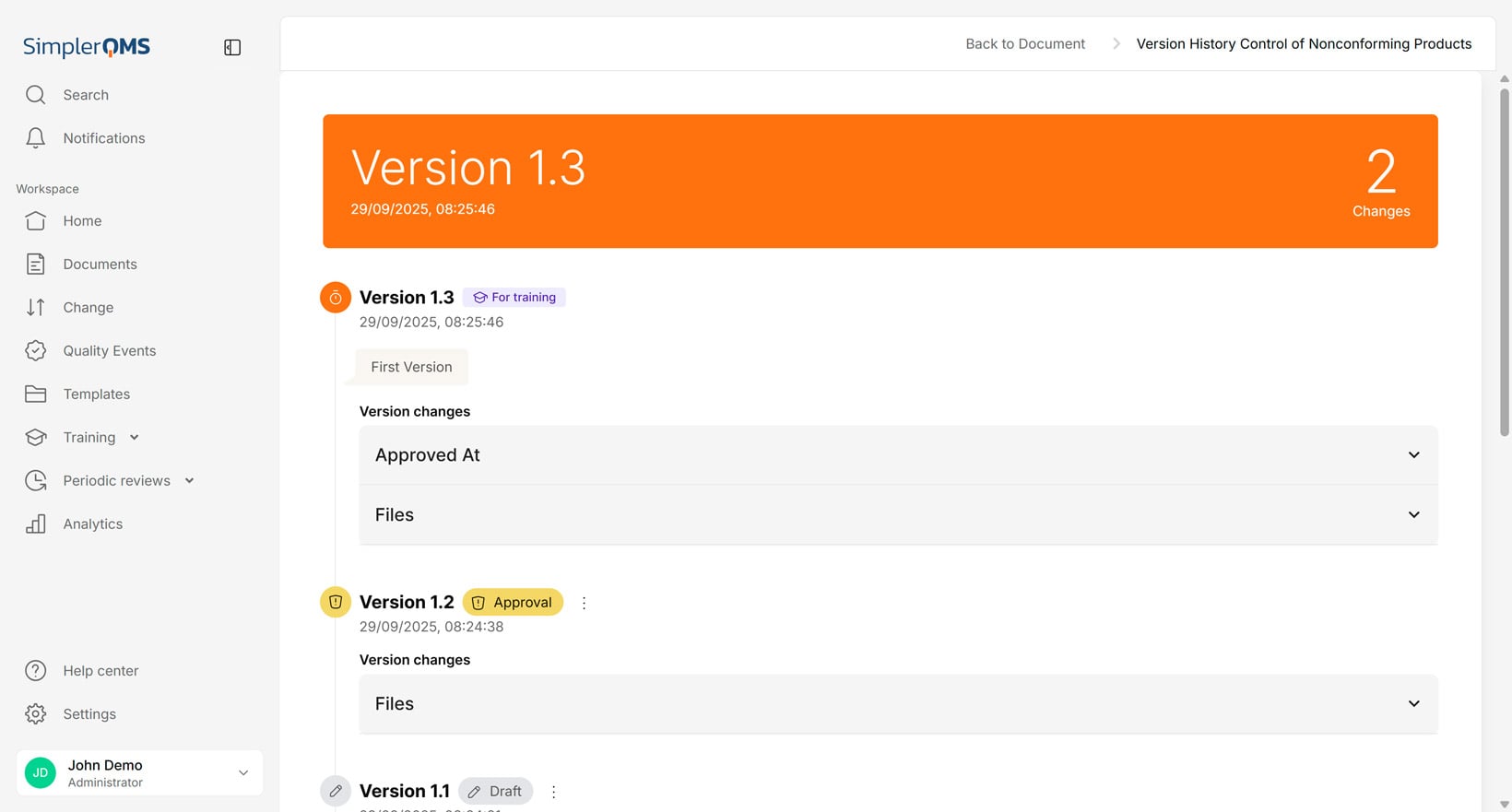
Audit Trail
The audit trail also referred to as a history file, is a recorded history of all changes made to a document. The audit trail includes information about who made the change when it was made, and what was altered.
The audit trail provides accountability and transparency, which are essential for compliance.
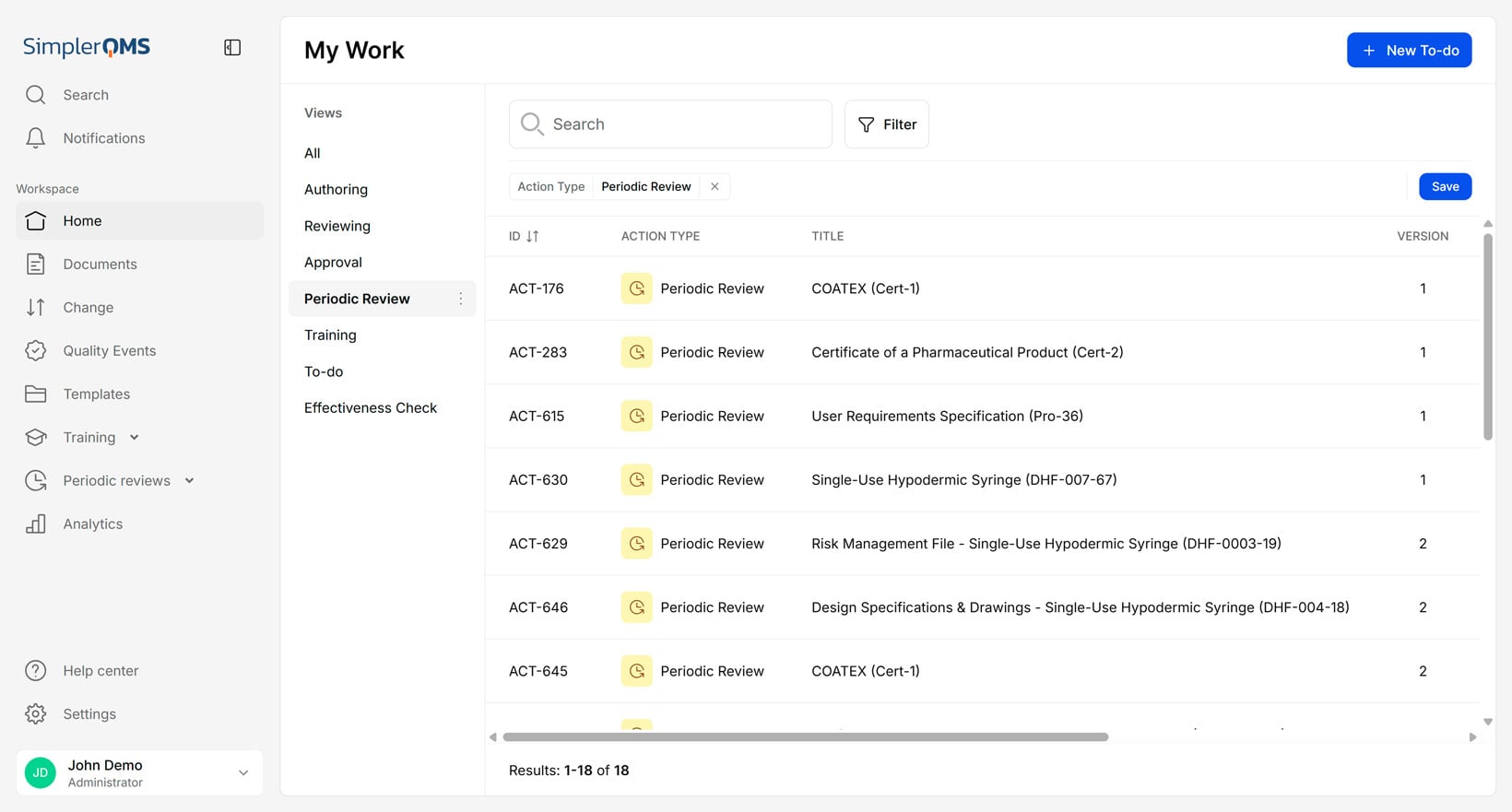
Periodic Review of Documents
Periodic review of documents is a scheduled process to review documents to ensure they are up-to-date and reflect current practices and requirements.
The periodic review of documents helps prevent the use of outdated documents and ensures compliance with relevant requirements. For example, every year, a company may conduct a periodic review of its SOPs to ensure they remain accurate, relevant, and compliant with internal specifications and requirements.
Access Control
Access control is the management of who can view, edit, or approve documents.
Access control protects information from unauthorized access or changes, ensuring that only qualified personnel handle documents.
Change Control
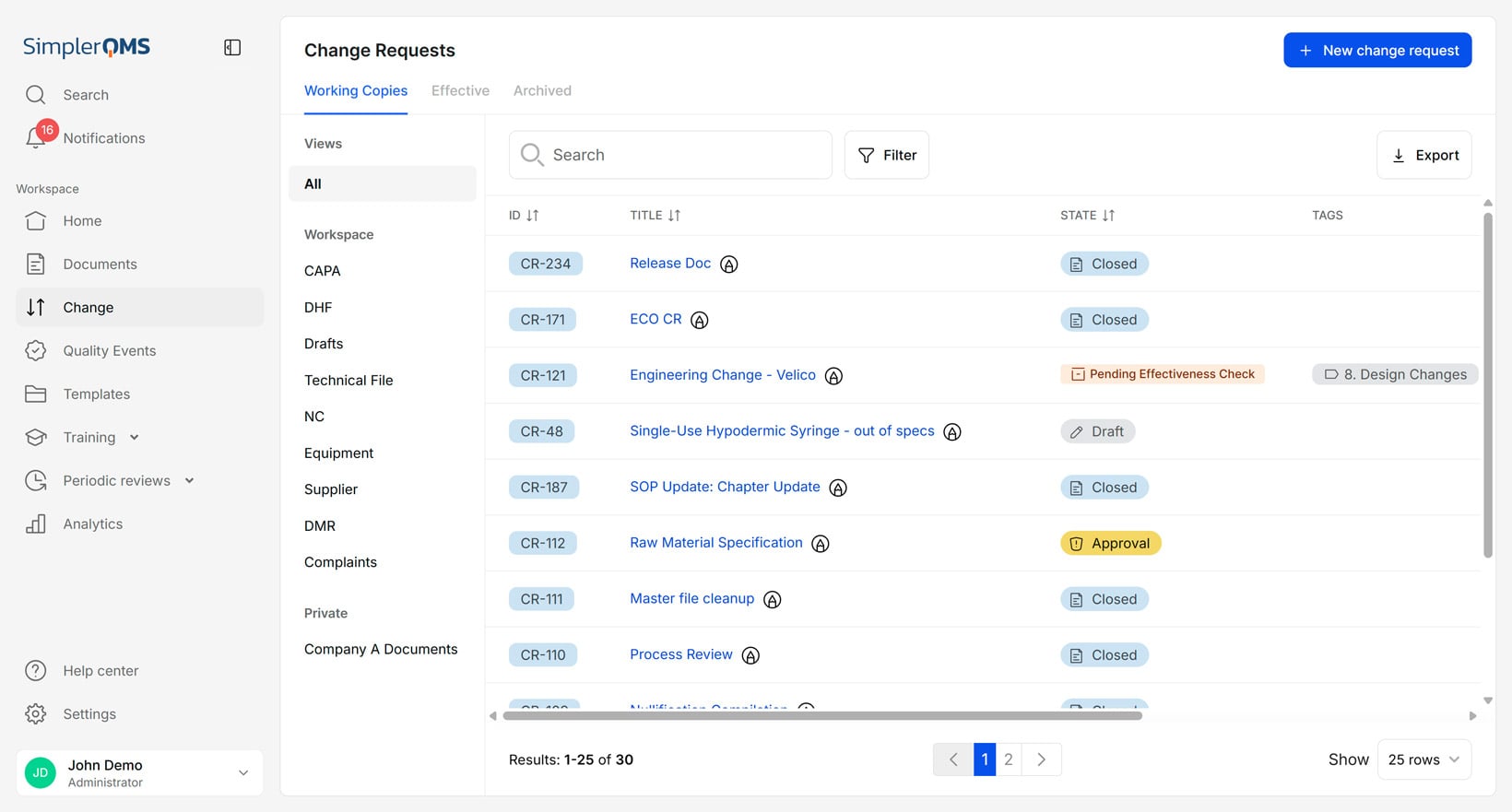
Change control within document control is the process of systematically controlling and documenting changes to documents and records.
Change control prevents unauthorized or untracked changes, ensuring that all document modifications are controlled, traceable, and compliant with relevant requirements.
Which Companies Could Benefit From a Document Control System?
Several types of companies can benefit from a document control system, such as the following.
- Medical Device
- Pharmaceutical
- Laboratory
- Healthcare
- Manufacturing
- Engineering and Construction
- Information Technology
- Legal and Professional Services
Medical Device
Companies in the medical device industry use document control systems to help ensure compliance with regulatory standards and regulations, such as ISO 13485:2016 and FDA 21 CFR Part 820.
Document control ensures all documents are managed and traceable throughout the design, development, and manufacturing of medical devices. Medical device document control includes procedures for handling documents such as design history files, device master records, validation protocols, and others.
Pharmaceutical
Companies in the pharmaceutical industry use document control systems to comply with regulations, standards, and guidelines such as GMP, FDA 21 CFR Part 211 and 212, ISO 9001:2015, and ICH Q10.
Document control ensures all documents are controlled and traceable throughout the drug development and manufacturing process. Pharmaceutical document control comprises procedures for controlling documents and records, such as batch records, SOPs, equipment validation protocols, and other documents.
Laboratory
Laboratories use document control systems to ensure transparency, traceability, and compliance with standards and regulations such as ISO 17025:2017, CLIA, and GLP.
Document control ensures all laboratory documents are controlled and traceable, ensuring the integrity of data and compliance with relevant requirements. Laboratory document control involves controlling documents such as procedures, test methods, calibration records, and others.
Healthcare
Healthcare companies use document control systems to comply with standards such as ISO 9001:2015.
Document control ensures that documents are secure and accessible. Healthcare document control includes managing patient records, research data, and compliance-related documentation.
Manufacturing
Manufacturing companies use document control systems to comply with standards and regulations such as ISO 9001:2015 and GMP requirements.
Document control ensures all production-related documents are controlled and traceable throughout the manufacturing process. Manufacturing document control includes controlling engineering drawings, SOPs, work instructions, equipment maintenance logs, and other documents.
Engineering and Construction
Engineering and construction companies use document control systems to control project documents and ensure stakeholders have access to the most current information.
Document control ensures that documents are properly controlled and accessible throughout the project lifecycle. Engineering and construction document control includes managing blueprints, project plans, contracts, safety documentation, and more.
Information Technology
Information technology (IT) companies use document control systems to manage compliance with standards such as ISO 27001:2022.
Document control ensures that documents are secure and traceable, including those related to data security and privacy. IT document control includes controlling policies, incident response plans, and compliance documentation.
Legal and Professional Services
Legal and professional services firms use document control systems to control client documents and comply with relevant requirements.
Document control ensures that legal and professional documents are secure and accessible only to authorized personnel. Legal document control includes managing contracts, client files, case documents, and compliance documentation.
What Are the Benefits of an Effective Document Control System?
The eight main benefits of an effective document control system are listed below.
- Ensures Regulatory Compliance: The document control system helps ensure regulatory compliance by controlling documents and records throughout their lifecycle as per requirements.
- Improves Quality Management: The document control system improves quality management by ensuring that all documents, such as SOPs, work instructions, and policies, are properly controlled and only the most recent version is utilized.
- Ensures Traceability: Document control systems improve traceability by tracking document versions, changes, and approvals, providing a clear audit trail.
- Increases Operational Efficiency: An effective document control system increases efficiency by eliminating administrative document-related tasks and allowing employees to focus on more value-adding tasks.
- Supports Collaboration: A document control system supports collaboration by allowing employees to access, review, and approve documents in a centralized and controlled system.
- Protects Data Integrity: Document control systems protect data integrity by restricting access, preventing unauthorized changes, and ensuring records remain secure.
- Supports Audit Readiness: A document control system simplifies audits by ensuring all required documents are easily accessible.
What Is an Electronic Document Management System (EDMS)?
An Electronic Document Management System (EDMS) or document management software is a software solution designed to manage, organize, and store digital documents in a centralized document repository. EDMS ensures secure access and efficient handling of documents throughout their lifecycle.
Basic systems, such as Dropbox, Google Drive, and SharePoint, focus on file storage and document sharing. Advanced EDMS solutions offer additional features such as document routing, electronic signatures, and predefined workflows to improve document management and support compliance.
Furthermore, advanced EDMS offers document control capabilities, allowing companies to manage and control documents and records. We will focus on advanced EDMS in the following sections.
What Are the Benefits of an Electronic Document Management System (EDMS)?
The seven main benefits of an electronic document management system are as follows.
- Helps Ensure Compliance: Helps companies comply with relevant requirements by providing document version control, audit trails, and predefined workflows.
- Increases Document Security: Provides robust security features, including access controls and encryption.
- Increases Efficiency: Automates document management processes, reducing manual tasks such as filing, versioning, routing, retrieving, and archiving documents.
- Improves Collaboration: Enables employees across departments to access and review documents, improving team collaboration.
- Reduces Costs: Minimizes the need for physical storage space, reduces document printing expenses, and lowers the costs related to managing paper documents.
- Secures Data: Ensures documents are backed up and can be recovered in the event of a disaster.
- Improves Accessibility: Provides quick and easy access to documents at any time from any authorized device.
What Are Examples of Electronic Document Management System (EDMS) Automation?
Examples of electronic document management system automation are described below.
- Document Routing: Automates the routing of documents to appropriate users for review and approval.
- Version Control: Manages and tracks document versions, ensuring only the latest version is in use. Document versions and changes can be tracked in the revision history for transparency and accountability.
- Compliant Electronic Signatures: Automates the process of adding timestamps and signature justifications when documents are signed electronically, ensuring compliance and traceability.
- Notifications and Reminders: Sends alerts for tasks, deadlines, and document updates.
- Audit Trails: Records all document actions for compliance purposes in a time-stamped audit trail.
- Access Control: Manages user permissions for document access and editing.
- Document Backup and Disaster Recovery: Creates and stores backups of documents in case of data loss or system failures.
Document Routing
The automation of document routing streamlines the workflows for reviewing and approving documents.
The automation speeds up the document routing by automatically moving documents through predefined workflows.
In SimplerQMS, documents are automatically routed to designated reviewers and approvers, ensuring that all necessary steps in these processes are followed.
The illustration below represents the steps in the document routing automation.
Version Control
Version control automation automatically assigns a version number to documents and tracks different versions. The version control automation ensures that users always have access to the most recent document version. It maintains a history of versions and related changes for reference and compliance purposes.
SimplerQMS automatically archives obsolete document versions while making the latest version available to users. SimplerQMS helps ensure compliance and eliminates the risk of working with outdated documents.
The illustration below represents the version control automation.
Compliant Electronic Signatures
Electronic signature automation streamlines and automates the application, tracking, and verification of electronic signatures on digital documents. The automation ensures compliance with regulatory requirements while increasing the speed and security of the document approval process.
SimplerQMS allows users to sign off documents electronically using electronic signatures compliant with FDA 21 CFR Part 11. Compliant electronic signatures accelerate the approval process and ensure that signatures are trustworthy and traceable.
A signed document with an electronic signature specifies the signer’s name, date, and time, as presented below.
Notifications and Reminders
Notification automation automatically sends alerts to users regarding important updates, actions, or changes within a system, ensuring awareness and timely response.
Reminder automation automatically sends follow-up messages to remind users about pending tasks or deadlines, aiming to ensure tasks are completed on time.
In SimplerQMS, automated notifications and reminders alert users of required actions, pending tasks, and when they need to review or approve a document, ensuring tasks are completed on time.
The illustration below represents notification automation.
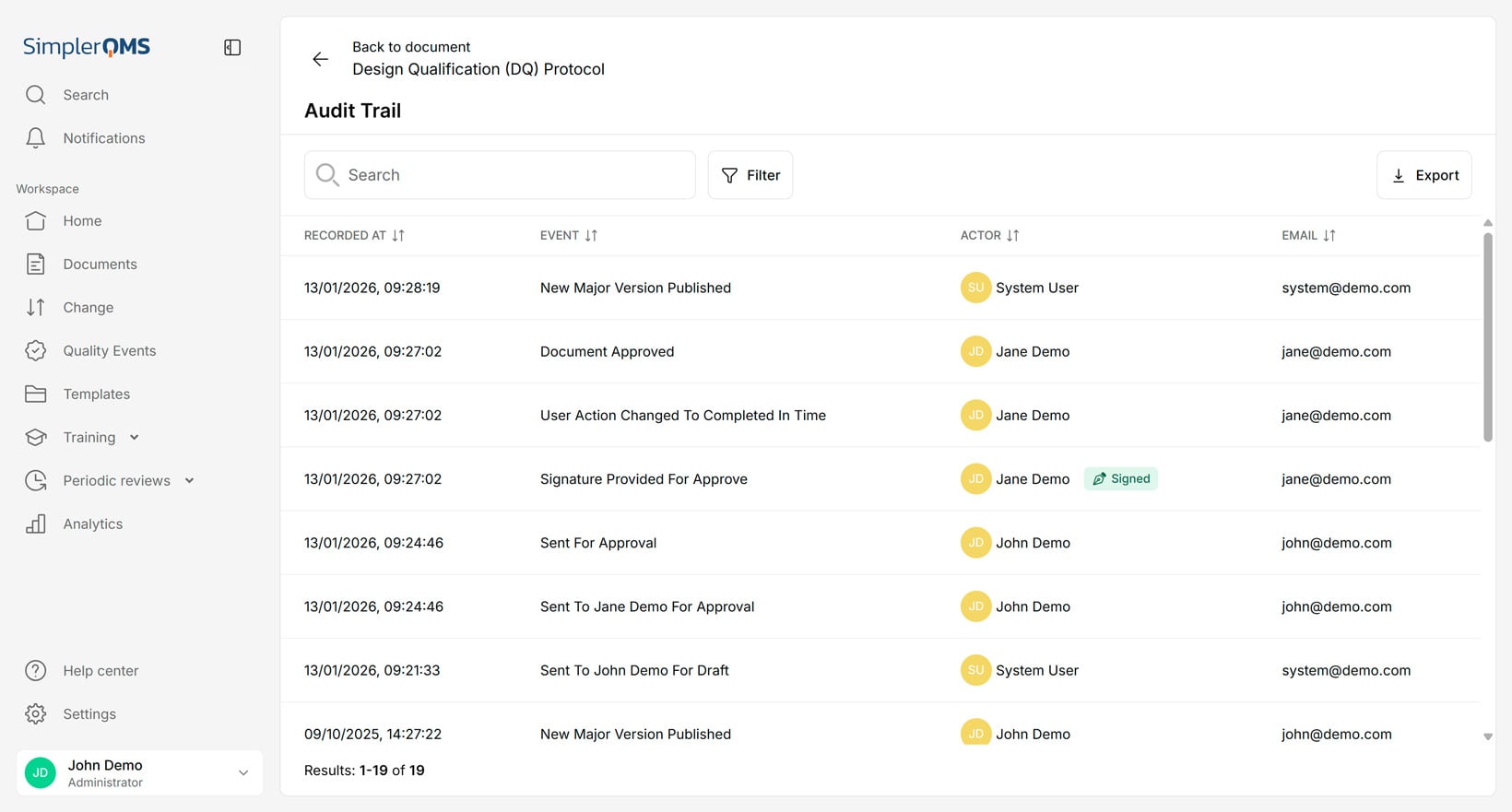
Audit Trails
Audit trail automation automatically records and maintains a detailed, time-stamped history of all user actions and document changes within the system. Audit trails, also referred to as history files, provide a traceable record for compliance and auditing purposes.
SimplerQMS automatically creates and stores time-stamped audit trails for each document, making it easy to track all activities and ensure compliance during audits. The audit trail is secure and cannot be tampered with or deleted.
The visual below illustrates an audit trail.
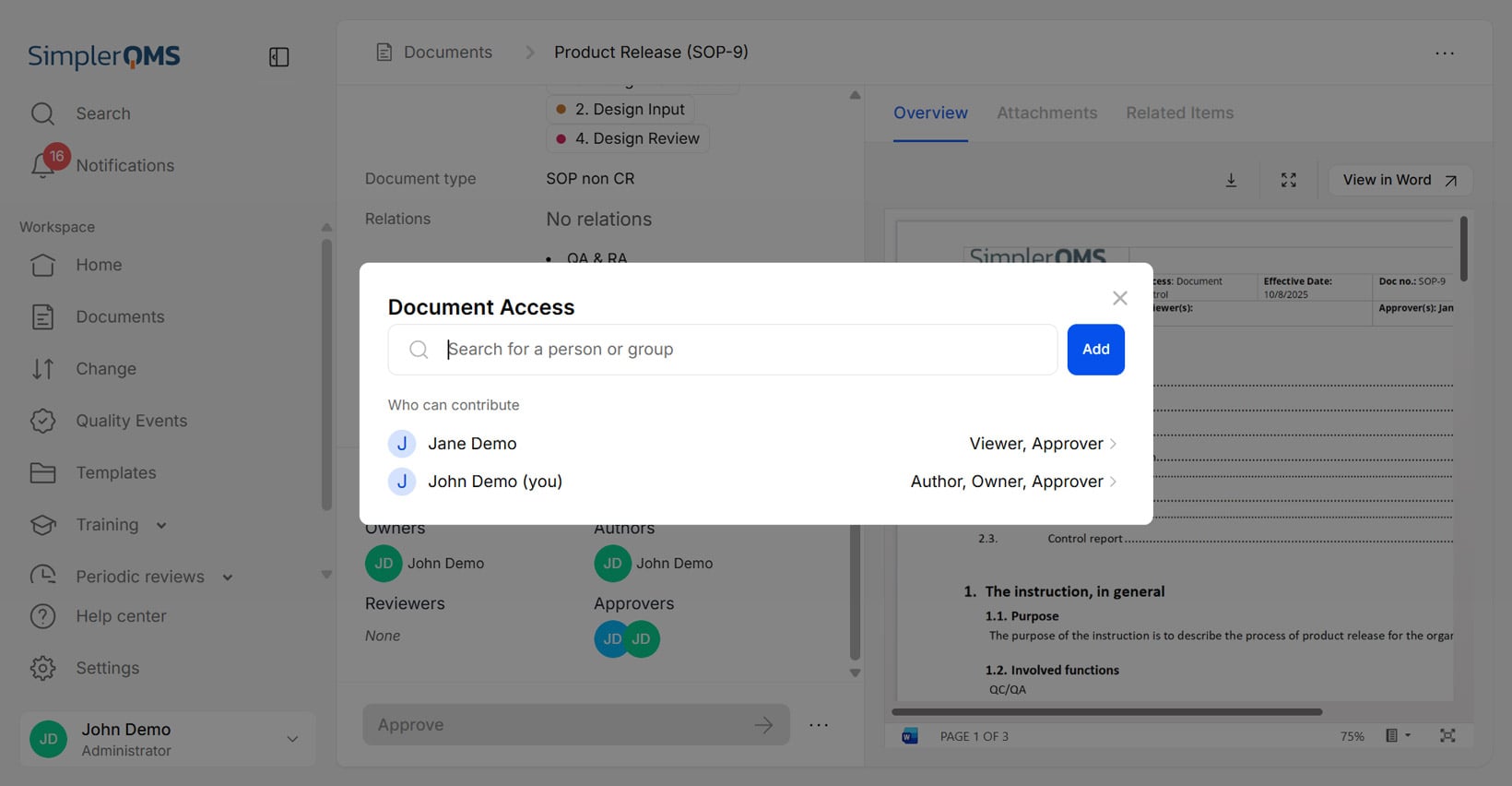
Access Control
Access control automation automatically assigns and manages user permissions within a system. Access controls ensure that only authorized individuals can access, change, approve, or dispose of specific documents based on their roles and responsibilities.
SimplerQMS allows administrators to set access levels for different employees based on their roles, sites, departments, or document types they need to handle. SimplerQMS access control capabilities ensure that only authorized personnel can access or modify documents.
The illustration below represents the document access control options.
Document Backup and Disaster Recovery
Document backup and disaster recovery automation is the process of automatically creating and storing copies of documents in the cloud at regular intervals to protect against data loss. The automation also ensures that documents can be quickly and reliably restored in the event of a system failure, data corruption, or other disasters.
SimplerQMS automates the creation of backups through Amazon Web Services (AWS), allowing for fast document recovery in the event of data loss and ensuring business continuity. SimplerQMS provides automatic cloud backups and AWS backup services every 15 minutes.

What Is the Role of Document Control in eQMS?
The role of document control in an eQMS is to handle document review, approval, tracking, and versioning to ensure document integrity, authenticity, and compliance with relevant requirements.
Document control ensures that only the latest approved versions of documents are accessible, tracking changes in a time-stamped audit trail. Document control in an electronic QMS helps ensure that all employees have access to the correct information when needed, supporting overall quality management and compliance.
SimplerQMS offers a life science eQMS solution with robust document control capabilities. SimplerQMS document control capabilities help ensure that all documents are managed efficiently and in compliance with relevant requirements.
Apart from document management and control, SimplerQMS provides comprehensive support for all QMS processes, including change control, training management, CAPA management, and audit management, among others. SimplerQMS offers predefined workflows that guide users through each process step, helping ensure compliance.
SimplerQMS supports compliance with QMS requirements, including the document control requirements outlined in ISO 9001:2015, ISO 13485:2016, FDA 21 CFR Part 820, ICH Q7, GMP, and others.
SimplerQMS is a fully validated solution in accordance with ISPE GAMP 5. The software validation process is entirely managed by SimplerQMS, saving customers from needing to invest resources and time in validation.
To see how SimplerQMS can streamline your document control and all the other quality management processes, book a personalized demo and talk to SimplerQMS Quality Solution experts.