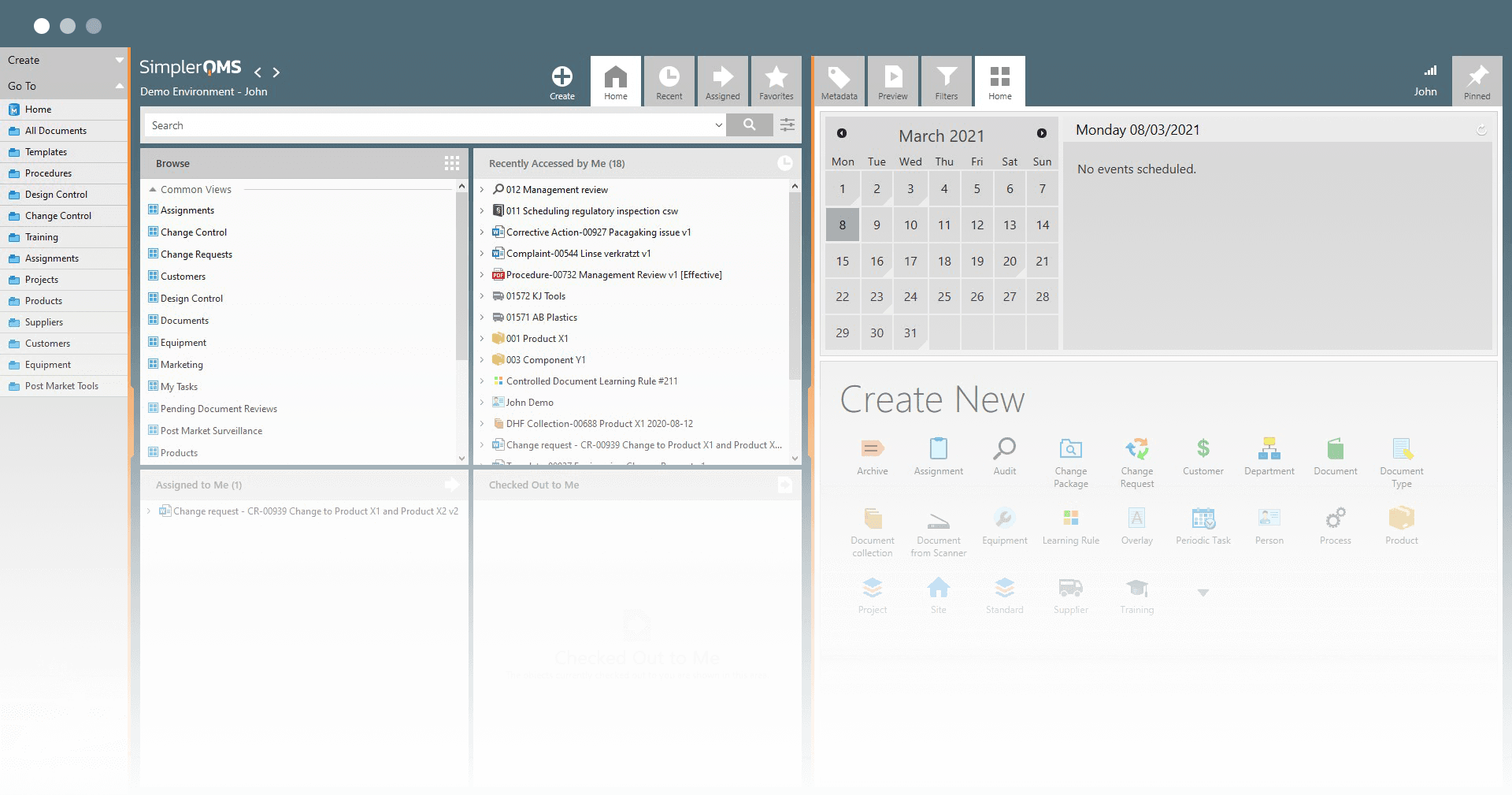
Get Rid of the Paper and Work More Efficiently
Discover how SimplerQMS will help your life science company save time and stay compliant.
TRUSTED BY
























SimplerQMS Offers an All-Inclusive eQMS Solution for the Life Science Industry
The SimplerQMS solution is all-inclusive, which means that you will not be charged for implementation, validation, user training, support, or hosting.
Everything is included and we don’t bill you by the hour. It’s that simple.
Avoid Time-Consuming Validation
We deliver a fully-validated eQMS in compliance with the regulatory requirements. We do all the software validation for you, eliminating any additional expenses, resources, or time commitments on your part.
Stay Compliant and Save Time
At SimplerQMS we take full regulatory responsibility for your eQMS allowing you to save resources and focus on your core business operations.
All eQMS Modules Included
All eQMS modules for life science companies are included – no matter if your company is in the early phase or if you have sales, production, and post-market activities.
Fast Implementation
Fast and effective implementation process that allows you to start reaping the benefits of eQMS software within just 5-6 weeks.
Unlimited Support and Guidance
We provide unlimited customer support and guidance to ensure that your eQMS is used efficiently and without interruption.
Work in Microsoft Office
SimplerQMS is seamlessly integrated into Microsoft Office. This allows your organization to work in familiar Office applications such as Word, Excel, and PowerPoint.
fully-validated software
Avoid Time-Consuming Validation
Computer System Validation is a requirement for life science companies that store and manage quality records digitally.
SimplerQMS provides a fully validated eQMS solution, which complies with the requirements of the GxP Guidelines, FDA 21 CFR Part 11, and ISO13485:2016.
This means that your organization does not have to spend any time or money on system validation processes. Furthermore, we ensure continuous re-validation of the software and provide extensive validation evidence for use during audits and inspections.
What kind of validation documentation and evidence do you provide?
- Validation Procedure
- Validation Plan
- Validation Report
- IQ, OQ and PQ Documentation
regulatory responsibility
Stay Compliant and Save Time
SimplerQMS takes full regulatory responsibility for your eQMS. Therefore, your organization does not need to invest in these specialized competencies.
We continuously monitor the Life Science regulatory requirements and update our processes and documentation accordingly.
Our customers are frequently audited by regulatory authorities and the SimplerQMS solution has been thoroughly examined with a successful outcome. We are ready to participate whenever your organization is audited or inspected – and SimplerQMS is always open for audits.
All regulatory documentation, such as validation reports and data retention policies, are available to your organization at any time.
Which standards and regulations does SimplerQMS comply with?
- FDA 21 CFR Part 11
- EU GMP Annex 11
- GxP Guidelines
- GAMP5
- ISO 27001
- ISO 9001:2015
- FDA 21 CFR Part 820
- ISO 13485:2016 (SimplerQMS is certified, as an organization, by Bureau Veritas)
- EU MDR & IVDR
- ICH Q10
- (and many more!)
modules
The SimplerQMS Modules Cover All Your Processes
No matter if your Life Science organization is a start-up or a later stage company, our integrated modules cover your needs. All of the modules are included in the SimplerQMS Subscription.
Training Management
Save time with automated training activities, learning overview, reminders, and generation of training certificates.
CAPA Management
Identify, uncover, resolve, and report all the preventative actions and corrective actions (CAPAs) seamlessly.
Complaint Management
Reduce the associated risks and resolve issues quickly by optimizing complaint management processes.
Change Management
Recognize and manage all changes accordingly to ensure compliance and structure within your organization’s QMS.
Design Control
Manage all the necessary processes related to product design and meet design control requirements with ease.
Document Control
Automate and standardize your document control activities with ease.
Risk Management
Consolidate risk and handle your risk management file in a well-organized and structured manner.
Supplier Management
Simplify supplier-related activities and handle your supplier documentation following the standards.
implementation
Establish Your eQMS in 5-6 Weeks
We know that your organization has limited time and resources for implementing new IT systems.
Therefore, SimplerQMS uses a fast and proven implementation method. The implementation typically takes 5-6 weeks.
The implementation process starts with the analysis of your needs and continues through configuration, training, migration, and an incremental launch of SimplerQMS modules.
secure cloud hosting
Secure and Scalable Hosting With Microsoft Azure
SimplerQMS uses a highly secure Microsoft Azure Cloud Storage solution. Your data is stored in state-of-the-art data centers managed by experts. This means that your organization does not need to invest in highly specialized competencies and expensive hardware.
Your users will be using the same password for logging in to Windows as for e-signing documents in SimplerQMS. This secure and user-friendly feature is included in SimplerQMS through the Microsoft Entra ID (previously known as Microsoft Azure Active Directory) technology.
With SimplerQMS you will have instant access to your documents – no matter where you are and what kind of device you are using. SimplerQMS can be used in any browser, through a Windows application, or by using our iOS or Android App.
platform and interface
Work in the Familiar Microsoft Office
SimplerQMS is seamlessly integrated into Microsoft Office. This allows your organization to work in familiar Office applications such as Word, Excel, and PowerPoint. Documents are simply edited in Office and saved with one click in SimplerQMS.
Our technology platform M-Files has integrations to Microsoft Office, Azure, Microsoft Dynamics, Salesforce, and many more applications. More than 5,000 companies worldwide are using the M-Files platform. This means that your organization will be using a proven technology platform while harvesting the benefits of SimplerQMS’ Life Science expertise.
support and guidance
Get Unlimited Support and Guidance
We understand the crucial role of the Quality Management System in your company. Efficient workflows should not be interrupted by troubleshooting or tutorial searching.
For that reason, we provide unlimited customer support. You can reach us by phone or e-mail when you need help. At the same time, SimplerQMS is regularly updated to ensure regulatory compliance, security and increased efficiency.
pre-configured eqms
Ready-To-Use to Use for Life Science
By using the ready-to-use SimplerQMS solution made specifically for Life Sciences, your organization will save time and money.
We configured all of the modules, so you can efficiently manage your SOPs, Instructions, Forms, Training Activities, Supplier Control, Technical File, Deviations, CAPAs, and much more.
At the same time, you get the flexibility to build dashboards (besides using our pre-defined dashboards), configure an unlimited number of projects, archives, and products, and store any file type of any size.
sops and templates
Save Time by Using Pre-Made Procedures and Templates
SimplerQMS includes a template package with a Quality Manual, Procedures, Forms, and Instructions based on the Life Science requirements such as ISO 9001, GxP, ISO 13485, FDA 21 CFR Part 210, 211, 820, ICH Q10, EU MDR and IVDR, ISO 15189, and more. These can be used as a starting point if you don’t already have a QMS in place.
Furthermore, you will save time by adapting key procedures such as Document Control, Supplier Control, Training & Learning which are tailored for companies that are using SimplerQMS. This means that you get “eQMS Compliant” procedures, which have been audited and inspected multiple times by the regulatory authorities.
Can I also include my own templates?
Yes! It is very easy to add your own templates. You can use your current templates, mix them with the pre-made templates or only rely on the included SimplerQMS templates. Whichever suits you best.
export of documents
Export Your Files Easily
You can easily export all your documents and files including a document index. Simply press the export button, and all the files will be transferred to your computer in an easily accessible structure.
We already backup your files for you. However, this allows you to do your own backups or send document packages to external people.
See SimplerQMS in Action
To see SimplerQMS in action and learn how you can make the most of it, request a personalized demo presentation.