QMS Software for the Life Science Industries
Streamline your quality processes with quality management software for Life Sciences – optimize workflows and stay compliant.
TRUSTED BY
























Improve Life Science Quality Processes With a Complete QMS Software
It is essential for Life Science companies to manage and control documents, training, audits, and other processes in compliance with requirements to ensure the safety and efficacy of their products or services.
SimplerQMS offers complete QMS software that supports various Life Science quality management processes such as document control, change management, training management, non-conformance and deviation management, CAPA management, supplier management, and many others.
SimplerQMS offers all Life Science QMS modules, is fully validated and pre-configured, developed with Life Science requirements in mind. The system supports compliance with requirements, such as GxP, ISO 13485:2016, FDA 21 CFR Part 820, FDA 21 CFR Part 11, EU GMP Annex 11, EU MDR and IVDR, and others.
Seamlessly Interlinked QMS Modules
By having interlinked quality management processes, you can create relations between documents and ensure seamless operations across all processes.
All QMS modules are integrated into one comprehensive quality management software designed for Life Science companies, be it a pharmaceutical, medical device, CRO, CMO, CDMO, or medical laboratory company.
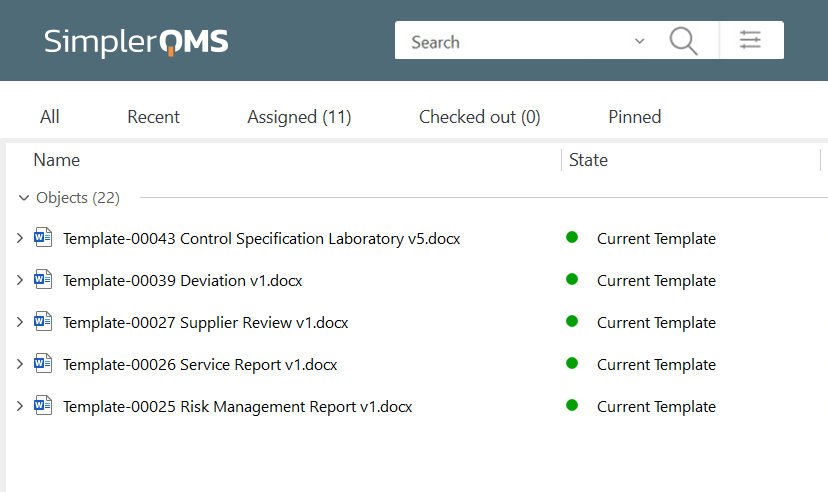
Effortlessly handle your procedures, document templates, and forms throughout all quality processes. You can also utilize our complimentary template package based on the Life Science requirements if needed.
Seamless Integration with Microsoft Office Applications
SimplerQMS offers seamless integration with Microsoft Office applications, allowing you to keep working in the familiar Word, Excel, PowerPoint, and Outlook applications.
Easily edit and save documents in the SimplerQMS cloud using just a few clicks.
Eliminate the need to download and upload files manually.
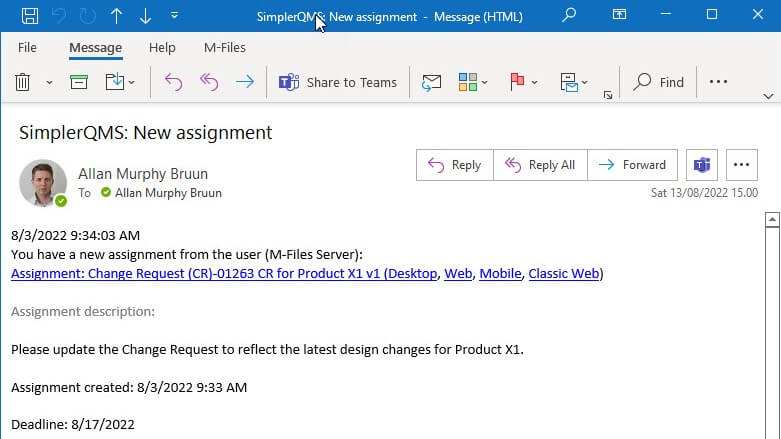
Automated Actions, Notifications, and Reminders
Define timelines when creating assignments and receive automatic reminders and notifications of any necessary actions before their due dates.
Have a complete time-stamped audit trail by automatically recording all actions performed in the system.
For example, receive automatic reminders when suppliers’ certificate expiration dates are approaching to ensure timely renewal. Ensure continuous training by sending automatic email notifications to relevant personnel regarding retraining in case of document updates, and more.
Fully Validated Quality Management Software
The system is fully validated according to ISPE GAMP5 to ensure it is developed, maintained consistently, and controlled following internationally recognized guidelines.
According to requirements such as FDA 21 CFR Part 11, EU GMP Annex 11, ISO, 13485:2016, and FDA 21 CFR Part 820, many Life Science companies are required to validate the software used in the quality management system for its intended use according to an established protocol.
We ensure that the software system is re-validated upon creating a new version or applying standard updates – allowing you to save both time and money on system validation processes.
Ensure Compliance with Life Science Requirements
SimplerQMS assists Life Science companies in achieving compliance with various industry requirements such as GxP guidelines, ICH guidelines, FDA regulations, ISO standards, and more.
Streamline processes using all Life Science QMS modules, pre-configured workflows, and, a complimentary template package.
SimplerQMS conforms to the guidelines of FDA 21 CFR Part 11 and EU GMP Annex 11 for electronic signatures, electronic records, and computerized systems in manufacturing.
We are also ready to assist you during your company’s audits or inspections and address any questions related to an eQMS.
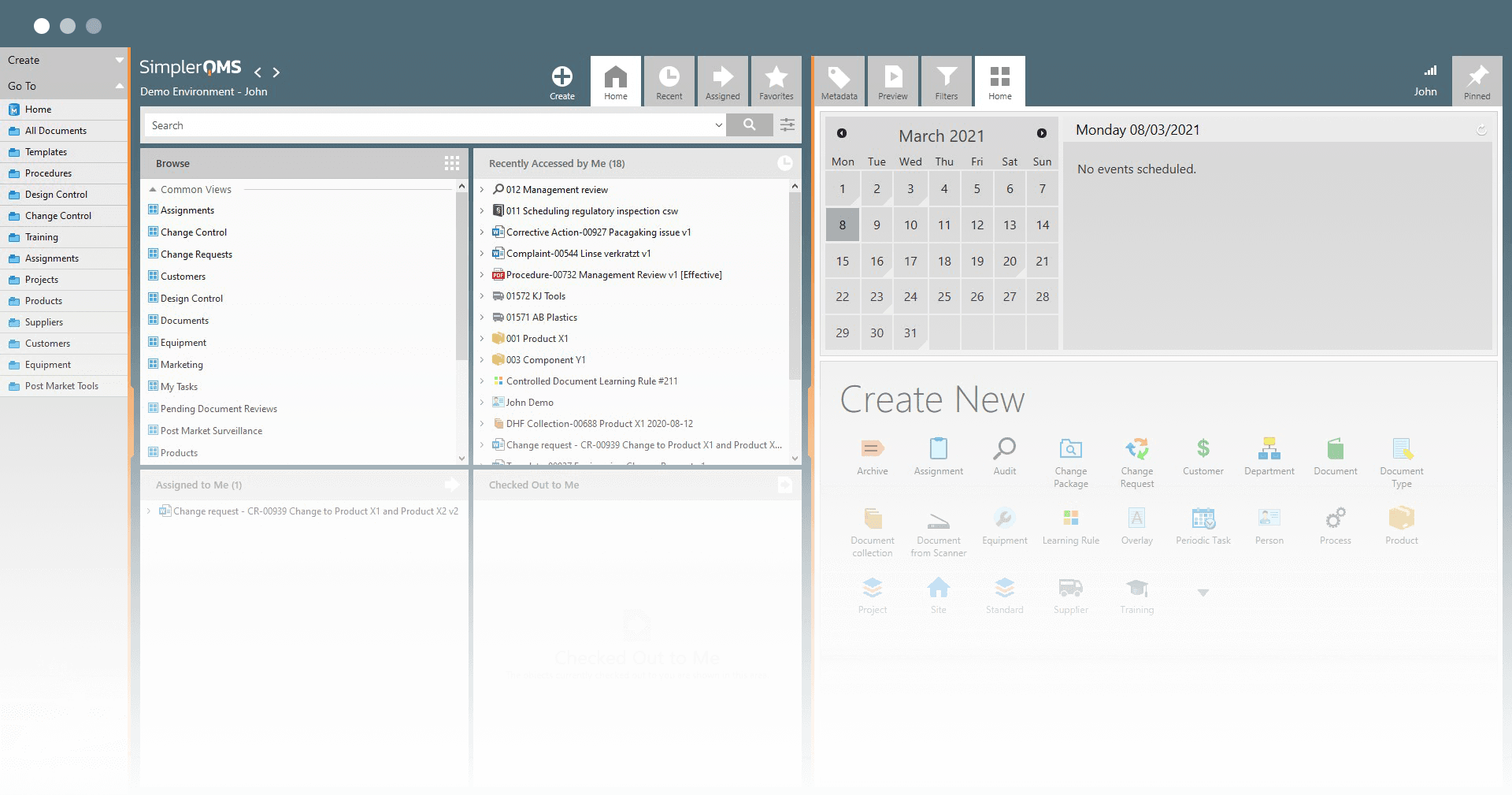
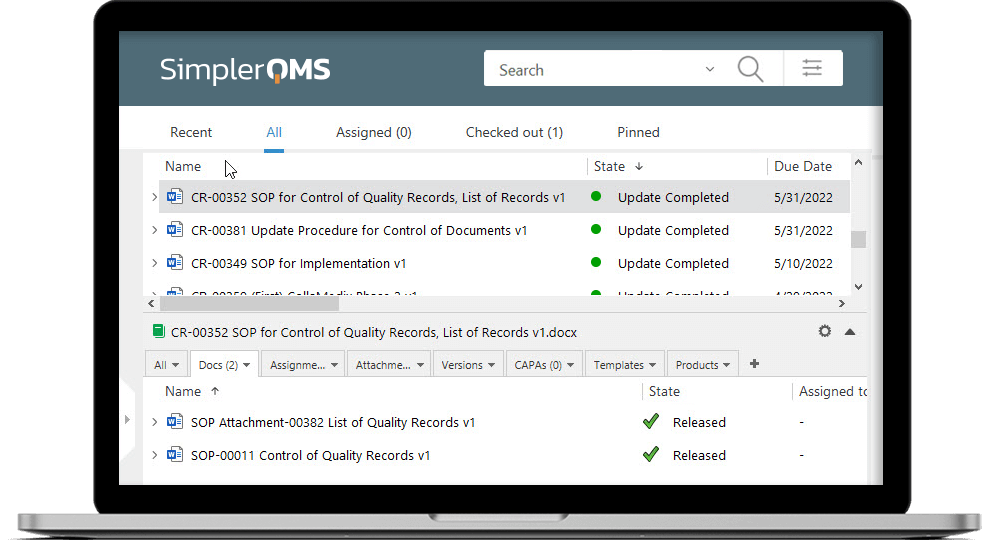
Maintain Document Control with Robust Features
SimplerQMS facilitates complex document control processes, including document approvals and version control.
Ensure easy access to documents using our cloud-based system, while maintaining document integrity by defining access level to documents.
Easily locate documents by searching for specific keywords within the title and content, simplifying the document retrieval process.
Efficiently Create Documents Using Document Templates
SimplerQMS offers a range of complimentary templates that can be used to create documents.
You can customize our templates to suit your specific needs, migrate your own templates, or create templates from scratch.
Utilize templates to create essential documents, such as SOPs, work instructions, non-conformance reports, CAPA forms, audit reports, etc., while working within familiar Microsoft Office applications like Word, Excel, and PowerPoint.
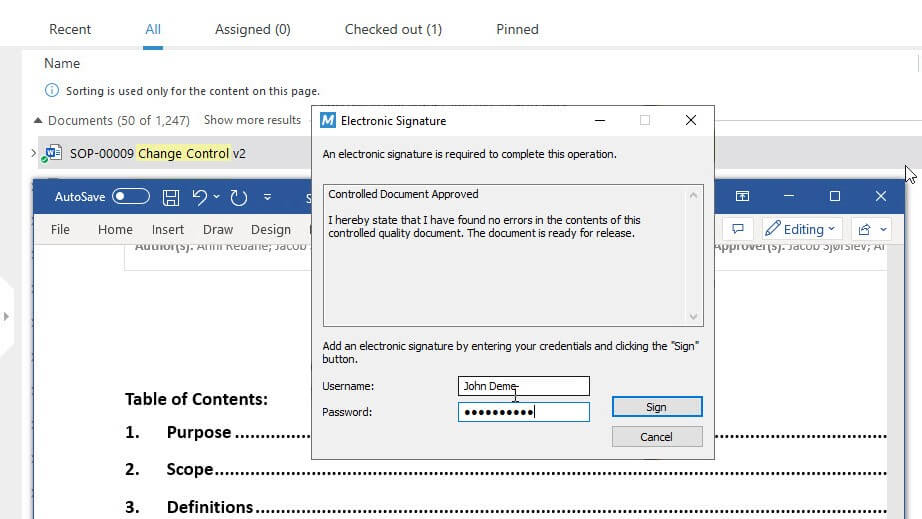
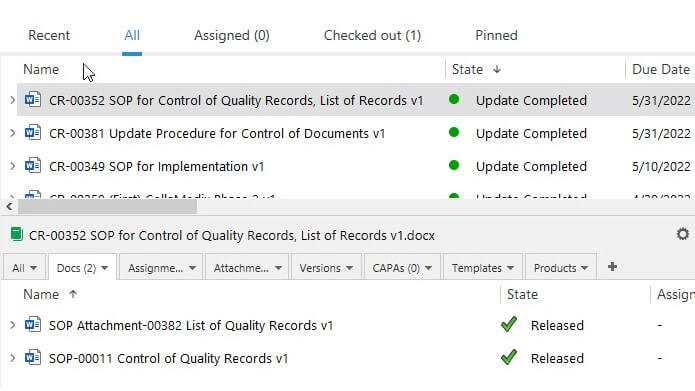
Control Changes with Robust Change Control Management
Address all steps involved in change control management such as creating change request documents using templates, routing change requests for review and approval, assigning specific tasks to individuals, and so on.
Electronically sign the change request to document the justification of the change and have all the steps automatically recorded in a time-stamped audit trail.
Ensure timely implementation of necessary actions by setting reminders for periodic reviews to check the effectiveness of a specific change.
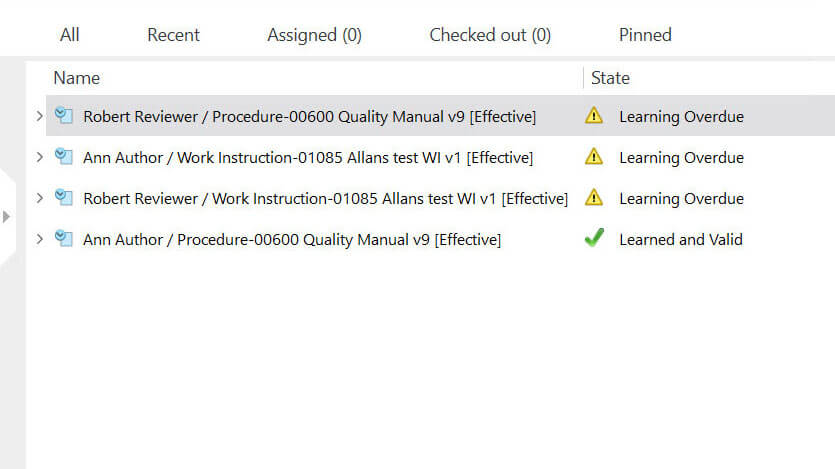
Streamline Employee Training Management
Create detailed training plans and designate learning to specific employee roles.
Automatically notify employees of upcoming training due dates or the need for retraining.
Keep track of each employee’s training progress and generate reports on their training completion status.
Maintain up-to-date and well-documented employee training records to ensure compliance with Life Science requirements.
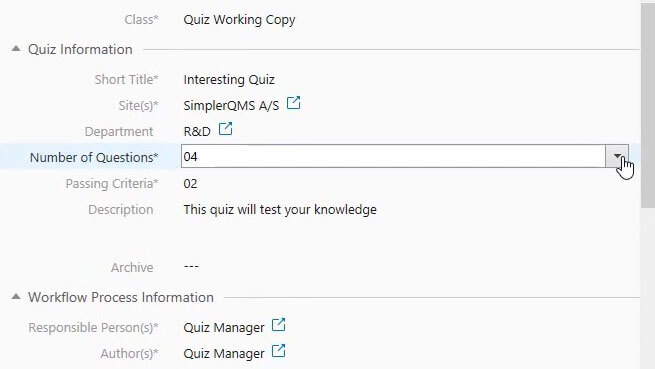
Ensure Effective Training with Quiz Assessments
Streamline the evaluation of training effectiveness by incorporating quizzes, and moving beyond just reading and signing documents.
SimplerQMS enables the measurement of training effectiveness through customizable quizzes presented in a multiple-choice format with pre-defined passing criteria.
Manage quizzes through the “draft, review, and approve” workflow before releasing and linking them to other learning documents.
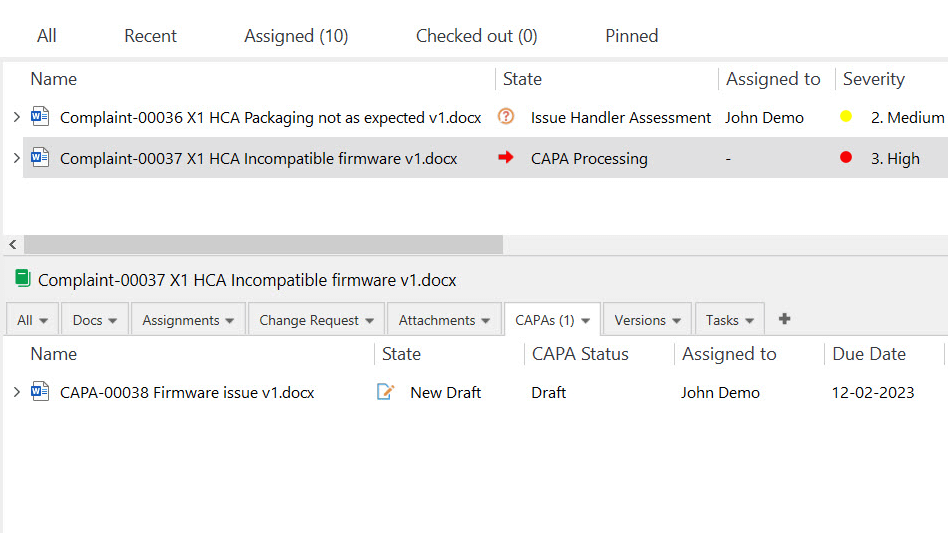
Track and Resolve Complaints Effortlessly
Capture and record all necessary information about the complaint. Route the complaint to the relevant personnel or department for investigation and resolution. Easily assign tasks and set deadlines to address complaints promptly and effectively.
Monitor each complaint’s status and comprehensively understand all complaint-related activities using highly customizable views.
SimplerQMS simplifies escalating complaints to the CAPA process to help investigate and address customer complaints properly.
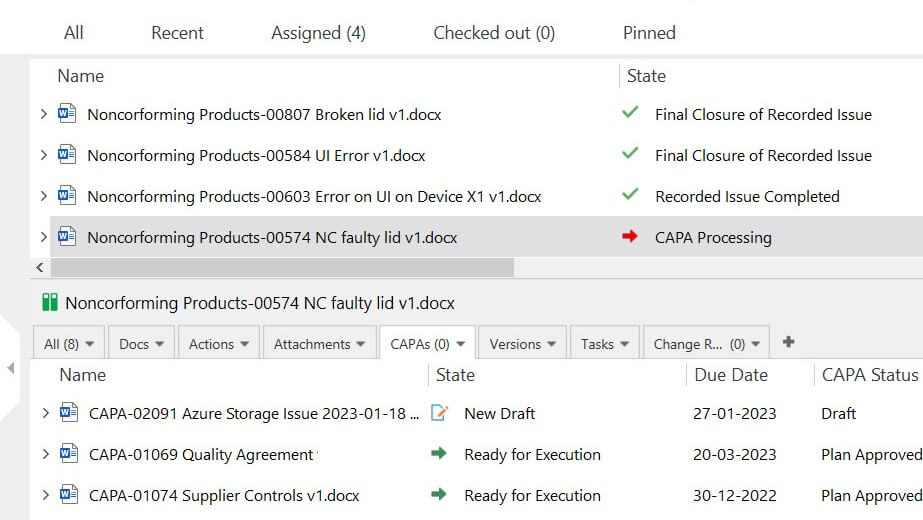
Efficiently Manage and Resolve NCs & Deviations
Handle quality events like non-conformances and deviations in a more efficient and organized manner.
Register the quality event, assign specific roles to the appropriate personnel, and route to review and approval.
Keep track of the status of every non-conformance and deviation via customizable views. You can also export data from these views to further analyze the trend over time.
Proactively Address Issues with CAPA Management
Easily escalate deviations and non-conformances, complaints, or audit findings to a CAPA.
Create a new CAPA document using a CAPA form template to ensure consistency and accuracy in the process. Create multiple CAPAs depending on the gravity of the issue.
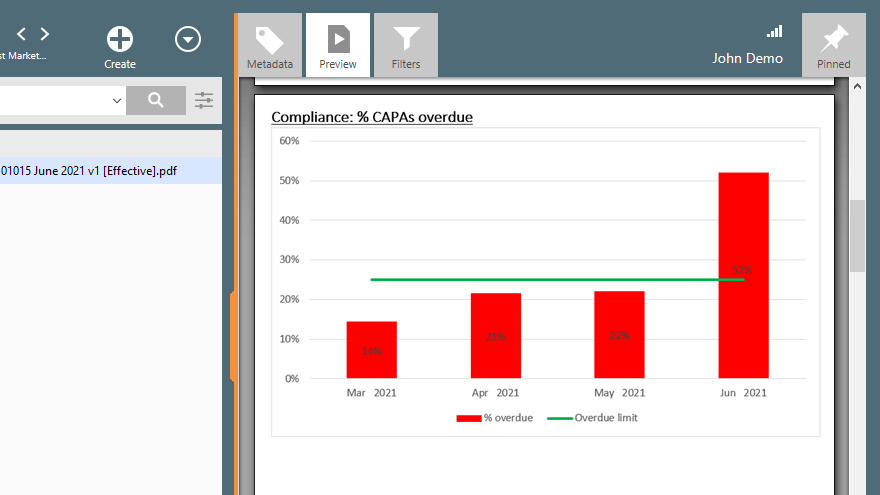
Check the effectiveness of a specific CAPA by setting reminders for periodic checks to ensure necessary actions are taken on time.
Export data and analyze CAPA trends to help identify improvement areas and implement preventive actions to address issues proactively.
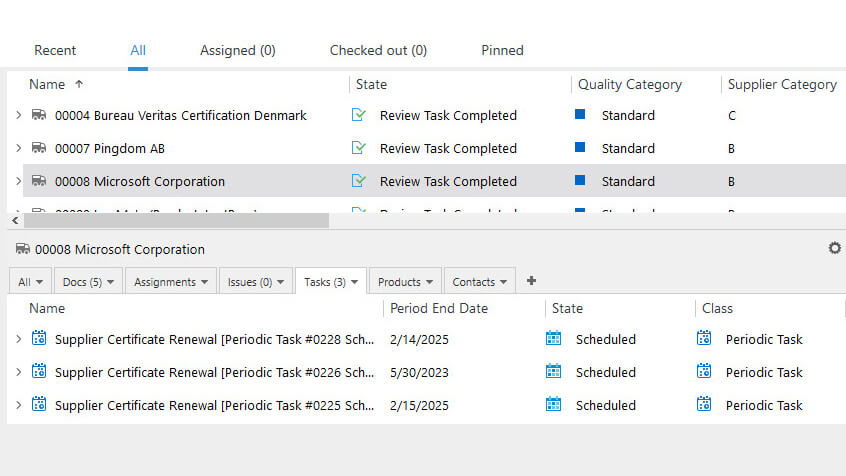
Effortlessly Manage Supplier-Related Processes
Manage all aspects of supplier management, including Approved Supplier Lists (ASLs), contracts, certificates, surveys, evaluations, supplier audits, and more, within a single system.
Simplify the management of supplier qualification, selection, and monitoring processes.
Easily schedule supplier requalifications, reviews, assignments, and other periodic or on-time tasks and maintain an overview of all relevant processes.
Establish appropriate access levels for supplier documents to allow for collaboration with external third parties.
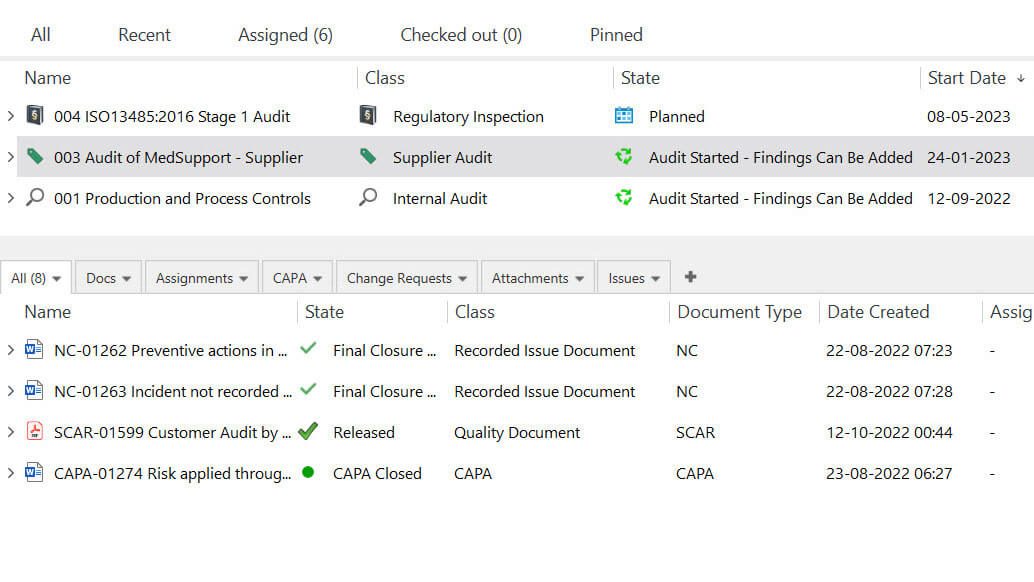
Assure Quality with Comprehensive Audit Management
Link individual audits to audit plans and keep track of all scheduled regulatory inspections, supplier audits, and internal audits.
Quickly retrieve documents using the search feature to provide relevant documents during audits.
Assign responsible persons to handle audit findings and quickly escalate audit findings to CAPAs if needed. Ensure complete traceability of information by linking audit documents to the corresponding CAPA, and much more.
Transparent Pricing for Maximum Budget Control
SimplerQMS offers all its modules and features under a single subscription price. The total cost of the subscription depends on the type and the number of licenses purchased.
All costs associated with implementation, hosting, system validation, user training, ongoing support, or additional modules are included in the subscription price of SimplerQMS. There are NO extra fees. Everything is covered – it’s that simple.
What Customers Achieve By Implementing SimplerQMS
Utilize Proven Technology
SimplerQMS is built on Microsoft & M-Files Technology which serves over 5,000 customers worldwide.
Pass Audit More Easily
Access needed documentation and present it to the auditor with a couple of clicks from anywhere in the world.
Gain High Level of Traceability
Gain cross-functional visibility and trace back to the root cause of each nonconformance.
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
Discover How SimplerQMS Can Help You
Complete Life Science eQMS Solution
Document Control
Streamline all document-related processes – document creation, routing, version control, audit trails, and more.
Change Management
Ensure structure and compliance within your company’s QMS by managing all changes appropriately.
Training Management
Create and execute complete training plans, automate notifications, and assess training effectiveness with quizzes.
Complaint Management
Manage complaints, monitor their status, and analyze the patterns and trends to identify improvement areas.
Risk Management
Manage your risk management files effectively and ensure compliance with Life Science requirements.
Equipment Management
Improve the management of equipment by easily controlling and managing equipment maintenance and calibration tasks.
CAPA Management
Seamlessly identify, uncover, resolve, and report all preventive and corrective actions.
Supplier Management
Streamline supplier-related activities and manage suppliers in accordance with specific Life Science requirements.
Audit Management
Improve audit management procedures and optimize the resources required to pass audits effectively.
Frequently Asked Questions
What Is Quality Management System Software?
Quality management system software is a digital tool designed to help companies implement and manage quality management processes. It is a comprehensive system that helps achieve consistency in processes, improve products and/or services, and enhance customer satisfaction.
A QMS software typically includes modules such as document management, corrective and preventive action (CAPA), change management, risk management, audit management, supplier management, training management, and others.
These tools help companies streamline their quality management system processes, monitor performance, and identify areas for improvement.
Which Are the Best QMS Software Solutions for Life Sciences?
Several QMS software solutions are specifically designed for Life Sciences companies operating in the pharmaceutical, medical device, biotechnological, and other Life Science industries. Each of them has its own characteristics and qualities.
When selecting a QMS software solution for Life Sciences, it is important to consider the specific needs of your company and the regulatory requirements you need to comply with. Choosing a software solution that is easy to use and provides good customer support to help with doubts and problems is also very important.
You can access our compiled list of the 6 best QMS software solutions for Life Sciences for a more straightforward evaluation.
You can also use our QMS software comparison template to compare the shortlisted QMS software against each other and select the best option for your company. Alternatively, you can utilize the template as a reference to develop your own quality management software requirements list.
What Types of QMS Software Exist?
Life Sciences companies require QMS software tailored to meet specific product types, industries, and market requirements.
Some examples of Life Sciences industries and solutions that support compliance with relevant requirements include the following.
Medical Devices
Quality management software for medical devices offers support for quality management processes that help ensure that medical devices are manufactured in compliance with quality standards and regulatory requirements. This solution helps with product realization, design, development, control of non-conforming products, and much more.
Pharmaceutical
Pharmaceutical QMS software solution helps pharma companies by providing the process support that helps ensure that their products comply with regulatory standards and are safe for human use. Such a solution can help manage pharmaceutical development, technology transfer, commercial manufacturing, product discontinuation, and so on.
Medical Laboratories
QMS solution for medical laboratories provides support for processes that help maintain accurate testing results and compliance with regulatory requirements. These solutions can help manage documentation, track quality control samples, monitor instrument performance, among, other things, and ensure adherence to relevant requirements.
What are Popular Quality Management Software Features?
Some popular Quality Management Software features are the following:
Document Control: enables users to create, store, and manage documents within a cloud-based system. It includes tools for version control, access control, and document tracking to ensure that documents are properly managed and tracked.
Change Control Management: allows users to manage document, process, and template changes within a controlled environment. It includes tools for change request management and pre-configured workflows to ensure that changes are appropriately evaluated, authorized, and implemented.
Training Management: includes tools for creating learning rules, managing training records, creating quizzes for evaluation of training effectiveness, automating notifications to remind employees of training, and more. These features help ensure that employees are adequately trained on quality-related processes, as well as improving quality and productivity.
CAPA Management: enables companies to escalate non-conformances and deviations, audit findings, or complaints to a CAPA and effectively manage corrective and preventive actions until closure while linking relevant documents to ensure traceability.
Audit Management: allows users to create audit plans, and schedule and link internal and external audits (including supplier and regulatory audits). It includes tools to manage audit plans, audit schedules, audit findings, audit reports, and more.
Besides these, QMS software vendors like SimplerQMS offer many more modules offering support for processes like Supplier Management, Non-Conformance Management, Complaint Management, and others. You can learn more about all the modules offered by SimplerQMS here.
How Much Does QMS Software by SimplerQMS Cost?
The pricing of SimplerQMS is determined by the type and quantity of licenses purchased.
SimplerQMS offers a subscription-based pricing model that includes all QMS modules, hosting, validation, implementation, user training, and ongoing support for one price, with no additional fees.
Please visit our pricing page to learn more about pricing and the features and services included.
How Much Time Does It Take to Implement SimplerQMS?
The implementation of the SimplerQMS solution typically takes between five to six weeks.
The duration of implementing the SimplerQMS solution can vary based on the size and complexity of your company, as well as the number of documents that need to be migrated into the system or created, and the available time resources.
Our experts will work together with you throughout and after the implementation process to guarantee a seamless and successful transition to quality management software.
See What Our Customers Have to Say
“Spending most of my day using SimplerQMS, I would say I am very pleased with the ease of use.”
Dorthe W.
QA/RA Manager, Cortex Technology
“SimplerQMS gave us excellent pricing, customer support for understanding how to use their system and set up our QMS, and is easy to use.”
Subba S.
Chief Technology Officer, CollaMedix
“Easy to work with. Intuitive. Rather easy to setup. Very good customer support. Good quality to price ratio.”
Jean Claude M.
Head of Hardware and Software Development, hemotune
See SimplerQMS in Action
To learn how you can make the most of SimplerQMS in your life science company – book a tailored demo.