Life Science Quality
Management Software
Comply with ISO, FDA, and GxP regulations and be audit-ready with our cloud-based quality management system.
Learn why life science organizations around the world trust SimplerQMS and achieve these results:
Full ISO, FDA, GxP, GMP compliance in audits
50% time reduction on change management
Increased efficiency and reduction in human error
Book a Demo
Join Over 5,000+ Quality Professionals Who Trust SimplerQMS
Work Efficiently in Microsoft Office Suite
Get your organization up and running fast on SimplerQMS.
We seamlessly integrate our eQMS with the familiar Microsoft Office applications – Word, Excel, Outlook, and PowerPoint.


Out-of-the-Box Regulatory Compliance
We provide an audit trail, versioning, and eSignatures, which are ISO, GxP, MDR, and FDA Compliant.
SimplerQMS is pre-validated QMS software and is ready for audits and inspections. Furthermore, we ensure continuous re-validation of the system.
Streamline Your Training Management Activities
Using SimplerQMS, you can create, assign, and track employee training tasks with ease.
Ensure that relevant employees receive training activities based on their functional roles.

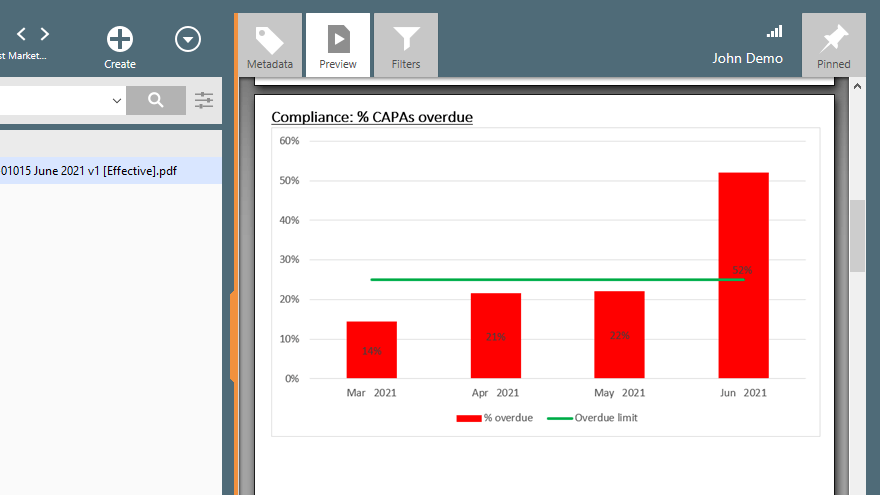
Overview KPI Reports
Get an overview of your organization’s training compliance, trending CAPAs, Non-Conformances, Deviations, and Complaints by viewing KPI reports.
Understand where your organization stands on critical quality metrics and historical trends.
Transparent, All-Inclusive Pricing
SimplerQMS is available as an all-inclusive annual subscription with flexible license options tailored to different roles and usage levels.
The subscription includes QMS modules, full system implementation, unlimited user training, system validation, hosting, QMS templates, and 24/7 support. There are NO extra fees. Everything is covered – it’s that simple.

One Platform That Connects All Quality Processes
Our quality management software integrates all your quality processes, and stores data in centralized, cloud storage, allowing you to manage risks, audits, suppliers, etc., in a single system.
From document control, with time-stamped audit trails and the FDA 21 CFR Part 11 compliant electronic signatures, to CAPAs or any other quality event, with automated assigning of tasks and notifications, SimplerQMS helps you work more efficiently and maintain compliance with ISO, FDA, and GxP regulations.
Document Control
Risk Management
Change Management
CAPA Management
Training Management
Design Control
Supplier Management
And much more…
What Customers Achieve With SimplerQMS
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
What Our Customers Say
“Spending most of my day using SimplerQMS, I would say I am very pleased with the ease of use.”
“SimplerQMS gave us excellent pricing, customer support for understanding how to use their system and set up our QMS, and is easy to use.”
“Easy to work with. Intuitive. Rather easy to setup. Very good customer support. Good quality to price ratio.”
See SimplerQMS in Action
To see SimplerQMS in action and learn how you can make the most of it, request a personalized demo presentation.
Book a Demo